Analytical Epidemiology — MCQs

A study that examines individuals who have already contracted a disease to identify risk factors is called:
In the context of a cohort study, which of the following statistical measures is not typically used?
Berksonian bias is a type of ?
According to Hill's criteria, which of the following is NOT a criterion for establishing causality in noncommunicable diseases?
The difference between the incidence in the exposed and non-exposed group is best given by:
A 10-year long study concerning the use of fluorides and pit and fissure sealants in the control of caries on the same population group is an example of:
What is the primary purpose of interventional studies in clinical research?
What is the definition of Population Attributable Risk?
A new test was developed for detection of COVID-19. What is the sensitivity of the test as per the information provided above?
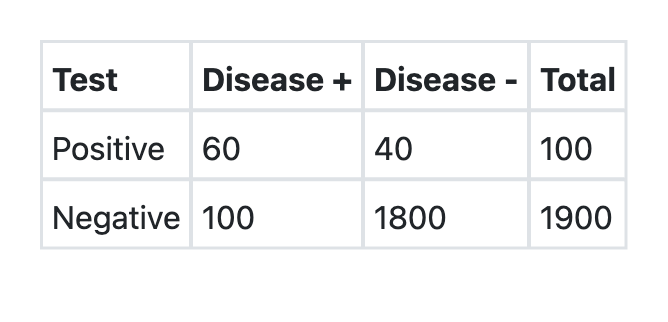
Which of the following Screening methods for Disease is the least useful?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
