Vitamins and Coenzymes — MCQs

On this page
Which is the water-soluble form of Vitamin K?
Vitamin K is involved in the post-translational modification of which of the following amino acids?
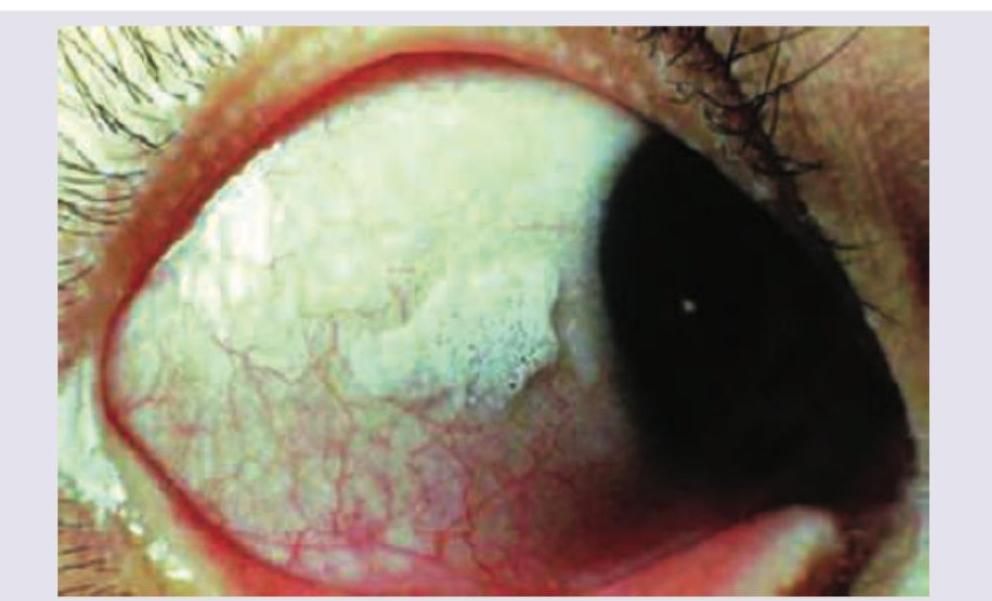
The condition shown in the image is caused by deficiency of which of the following vitamins?
The function of vitamin K largely depends on which mineral?
In which part of the intestine does vitamin B12 bind with intrinsic factor?
Decreased serum Vitamin B6 levels are associated with which of the following conditions or treatments?
Which of the following vitamins can act without phosphorylation?
Vitamin B6 is required for which reaction?
Hypervitaminosis of which of the following vitamins will cause bony abnormalities?
All of the following are characteristics of rickets except?
Practice by Chapter
Fat-Soluble Vitamins: A, D, E, K
Practice Questions
Vitamin A and Vision
Practice Questions
Vitamin D and Calcium Metabolism
Practice Questions
Vitamin E and Antioxidant Functions
Practice Questions
Vitamin K and Blood Coagulation
Practice Questions
Water-Soluble Vitamins: B Complex and C
Practice Questions
Thiamine (B1) and Pyruvate Dehydrogenase
Practice Questions
Riboflavin (B2) and Flavin Coenzymes
Practice Questions
Niacin and NAD/NADP
Practice Questions
Vitamin B6 and Transamination
Practice Questions
Folate and Vitamin B12 in One-Carbon Metabolism
Practice Questions
Vitamin C and Collagen Synthesis
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
