Post-Translational Modifications — MCQs

Which of the following statements regarding collagen synthesis is incorrect?
Ubiquitin is involved in what process?
Carboxylation of clotting factors by vitamin K is required for them to be biologically active. Which amino acid is carboxylated?
What is the major site of protein glycosylation?
Which of the following is not a characteristic feature of Alzheimer's disease?
Which of the following is considered a poor prognostic marker in multiple myeloma (MM)?
Statement 1 - A 59-year-old patient presents with flaccid bullae. Histopathology shows a suprabasal acantholytic split. Statement 2 - The row of tombstones appearance is diagnostic of Pemphigus vulgaris.
During eukaryotic protein synthesis, phosphorylation of which of the following is enhanced by insulin?
Protein segregation occurs in which organelle?
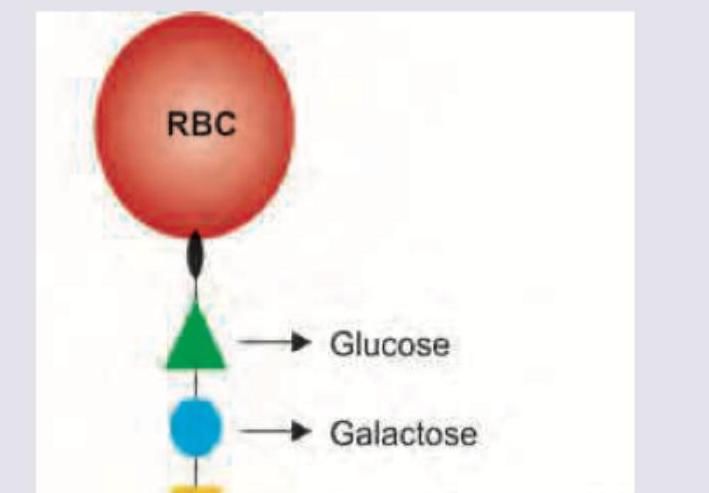
Name the antigen marked as X determining blood group A.
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
