Protein Structure and Function — MCQs

On this page
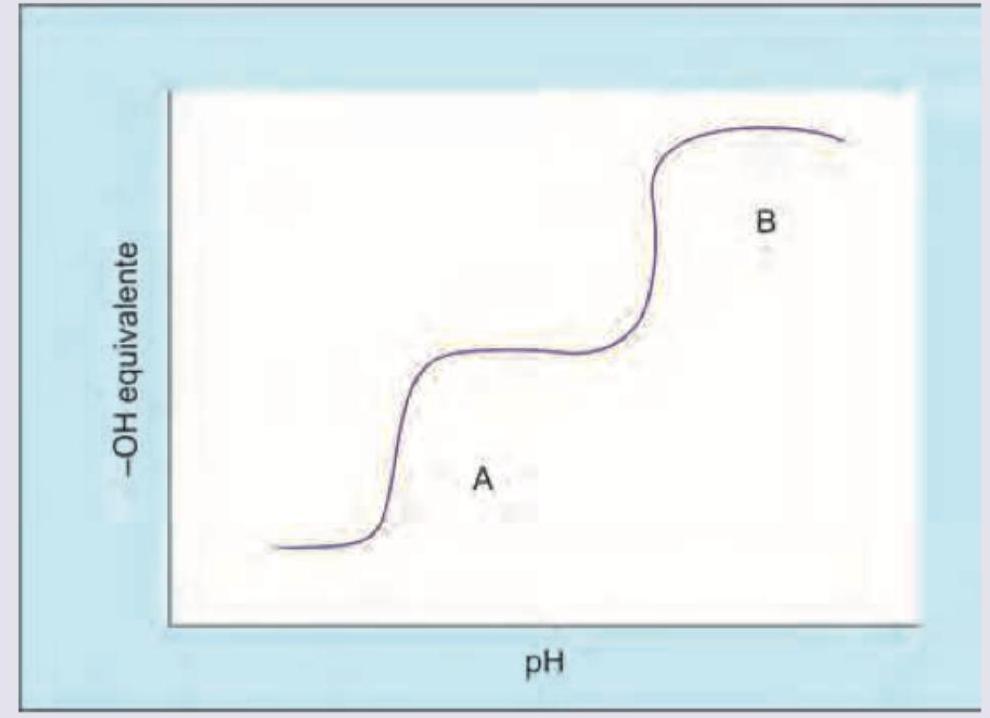
What is the pH of the zwitterion as shown in the titration curve of glycine?
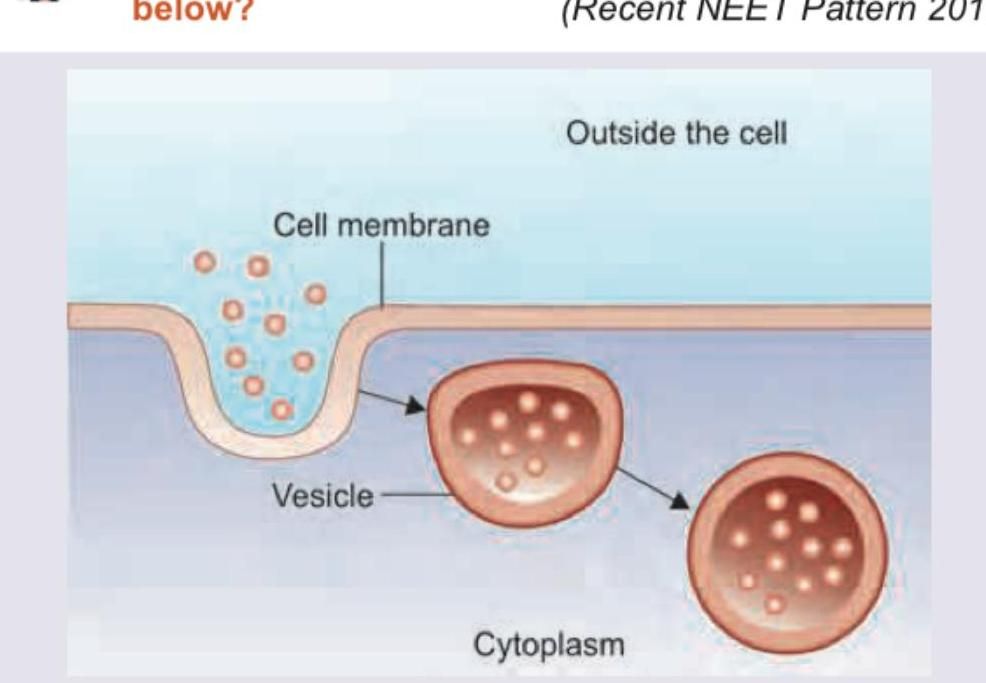
Which protein plays a role in the process shown below? (Recent NEET Pattern 2018)
All are correct about the protein shown in the figure except? (Recent NEET Pattern 2016-17)

All of the statements are true about the titration curve of a protein except:
Egg proteins are considered to be the best among food proteins because : I. of their biological value II. of their digestibility III. they contain all the essential amino acids IV. their calorie content is higher than in proteins of vegetable sources Which of the statements given above is/are correct?
The biological value of a given protein depends upon
Which one of the following statements about Human Immunoglobulins is not correct?
Compared to breast milk, colostrum has
Beta hCG is structurally similar to which biochemical moiety?
Chaperones are:
Practice by Chapter
Amino Acids: Structure and Properties
Practice Questions
Peptide Bond Formation
Practice Questions
Primary Structure of Proteins
Practice Questions
Secondary Structure of Proteins
Practice Questions
Tertiary and Quaternary Structures
Practice Questions
Protein Folding and Chaperones
Practice Questions
Protein Domains and Motifs
Practice Questions
Structure-Function Relationships
Practice Questions
Hemoglobin and Myoglobin
Practice Questions
Collagen and Elastin
Practice Questions
Albumin and Plasma Proteins
Practice Questions
Post-Translational Modifications
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
