Protein Structure and Function — MCQs

On this page
Which of the following is a denaturing substance?
Defects in protein folding result in which of the following clinical diseases?
What is the biochemical classification of gonadotropins?
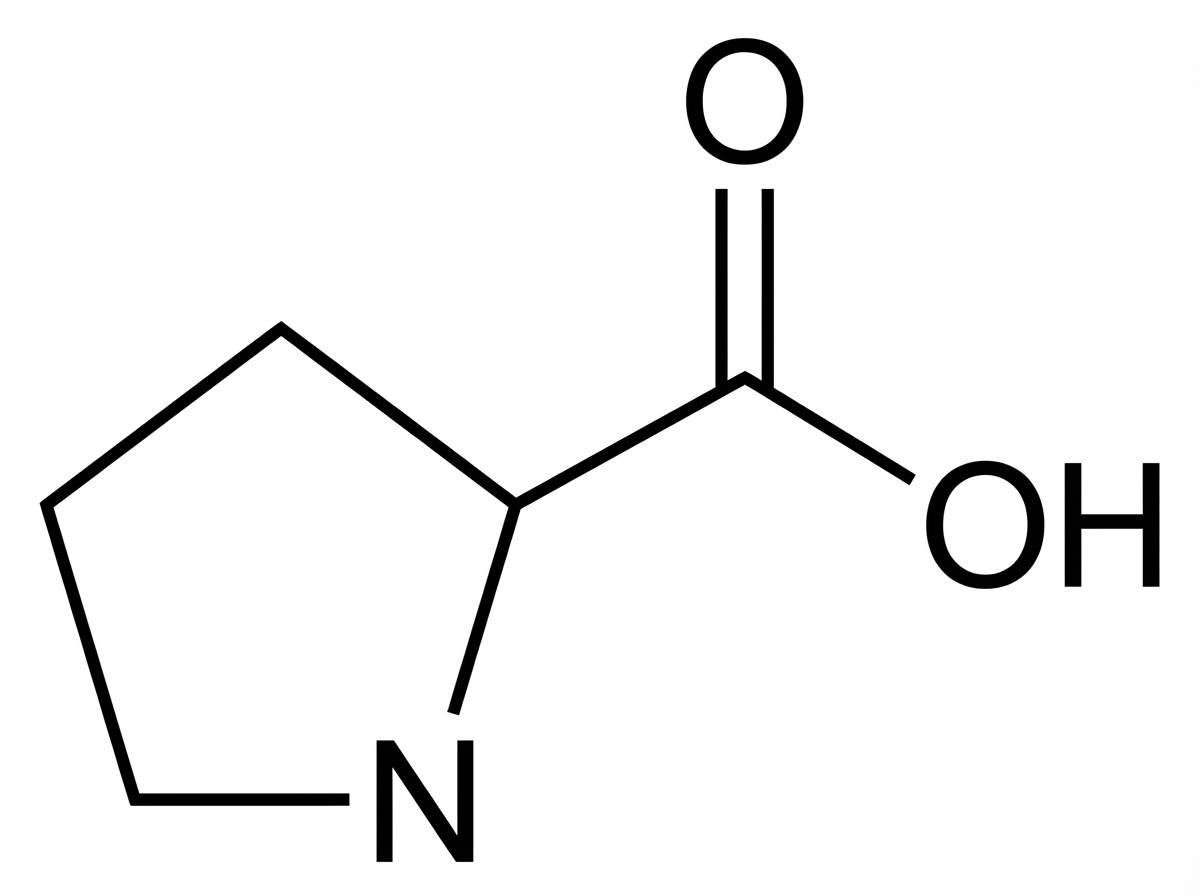
Which is the amino acid shown below?
Which of the following protein structures is not affected by denaturation?
Which of the following is an optically inactive amino acid?
In determining protein structure, what is the role of mercaptoethanol?
Which biochemical test requires an intact peptide bond for a positive result?
Which of the following is NOT a property of the Signal Recognition Particle (SRP)?
All of the following are required for hydroxylation of proline residues except?
Practice by Chapter
Amino Acids: Structure and Properties
Practice Questions
Peptide Bond Formation
Practice Questions
Primary Structure of Proteins
Practice Questions
Secondary Structure of Proteins
Practice Questions
Tertiary and Quaternary Structures
Practice Questions
Protein Folding and Chaperones
Practice Questions
Protein Domains and Motifs
Practice Questions
Structure-Function Relationships
Practice Questions
Hemoglobin and Myoglobin
Practice Questions
Collagen and Elastin
Practice Questions
Albumin and Plasma Proteins
Practice Questions
Post-Translational Modifications
Practice Questions
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
