Purine Metabolism and Disorders — MCQs

For the conversion of aspartate to asparagine, nitrogen comes from which source?
Mechanism of action of allopurinol is
Which of the following increases uric acid excretion?
A 5-year-old presents with intellectual disability, self-mutilation, and hyperuricemia. What enzyme defect is most likely?
A patient presents with hyperuricemia and gout. Which enzyme's overactivity is most likely associated?
Which molecule provides nitrogen-9 to the purine ring?
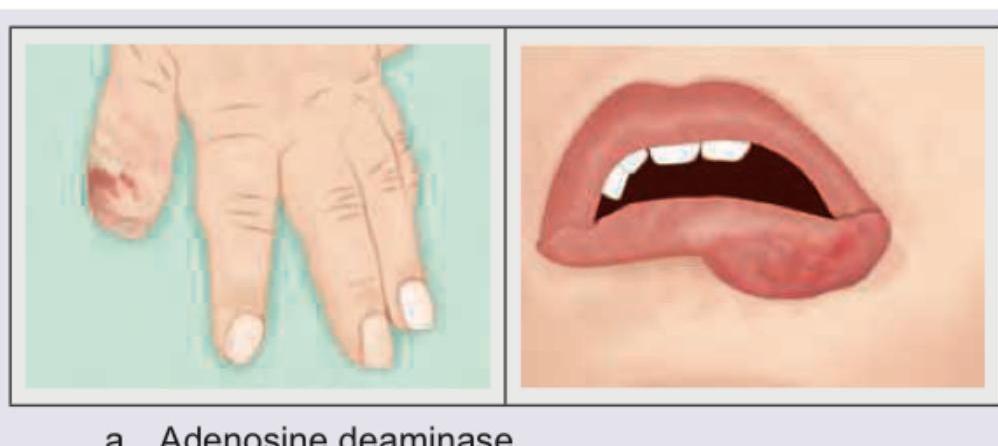
The patient shown here is suffering from deficiency of which enzyme?
Inosinic acid is biological precursor of ?
What is mechanism of action of colchicine in acute gout?
Which of the following organs does not primarily utilize the salvage pathway of purine nucleotide synthesis?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
