Chromatography Techniques — MCQs

Which of the following separates proteins solely on the basis of their molecular size?
Which method is used to separate a mixture of lipids?
The most specific test to detect blood stains is:
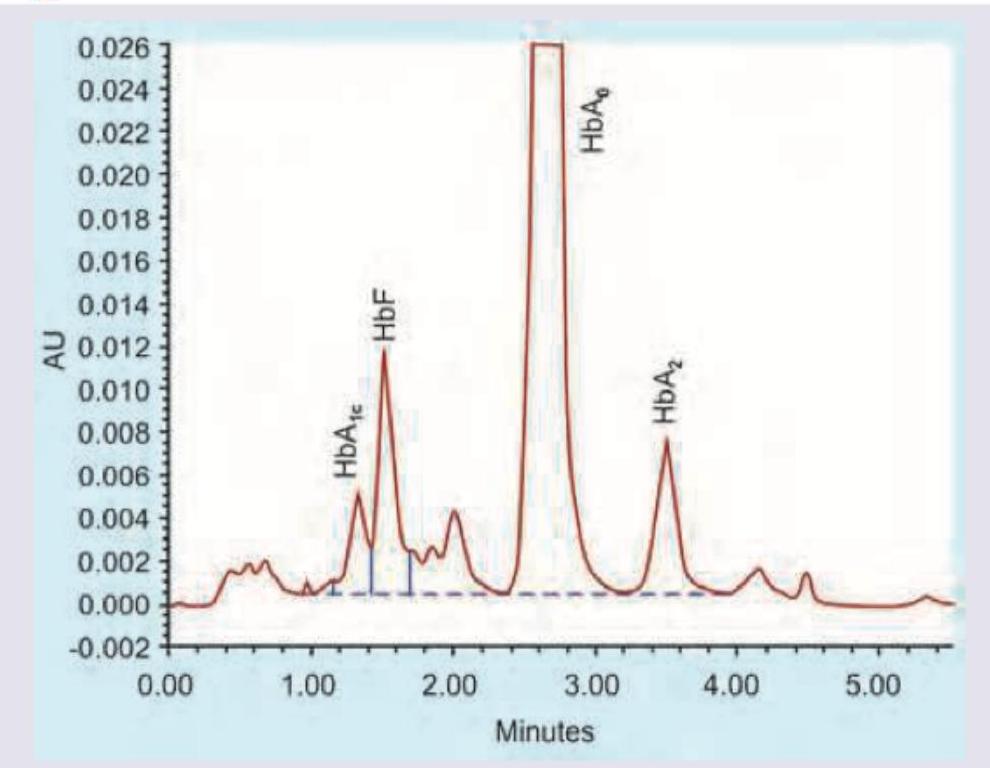
Glycated hemoglobin (HbA1c) is best measured using?
The technique shown in the image is:
Protein purification and separation can be done by all except:
Which technique is used for protein separation based on molecular size?
Expression and release of a repressed emotion is called as
Which of the following methods of protein separation is not dependent on molecular size?
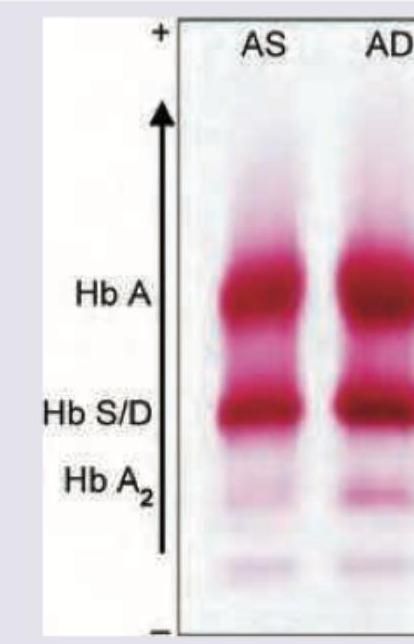
The shown pattern in electrophoresis is due to:
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
