Molecular Aspects of Development — MCQs

Sumoylation of histone proteins is associated with
Phenotypic expression of a gene depending on the parent of origin is referred to as:
The following gene mutation protects tumor cells from apoptosis:
Gene not involved in SCID:
Which of the following protein molecules is responsible for cell-to-cell adhesion?
Which of the following is not a part of extracellular matrix (ECM)?
At what week of gestation do limb buds appear?
The following ocular structure is not derived from surface ectoderm –
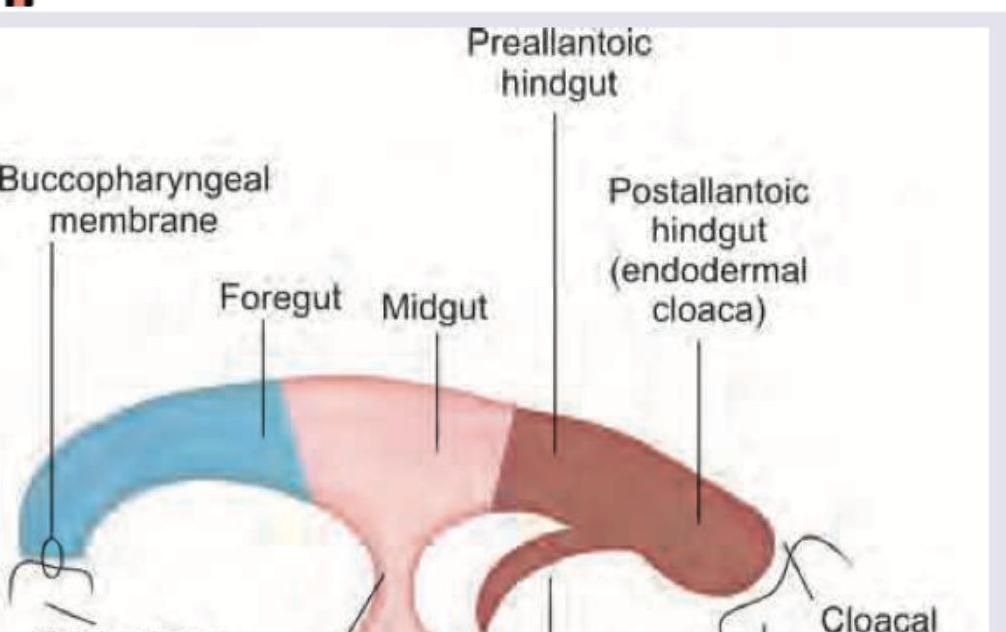
Which is wrong about the image given below?
Which paranasal sinuses are present at birth?
Want unlimited practice?
Get full access to all questions, explanations, and performance tracking.
Scan to download app
