HCV & DAAs - The Antiviral Arsenal
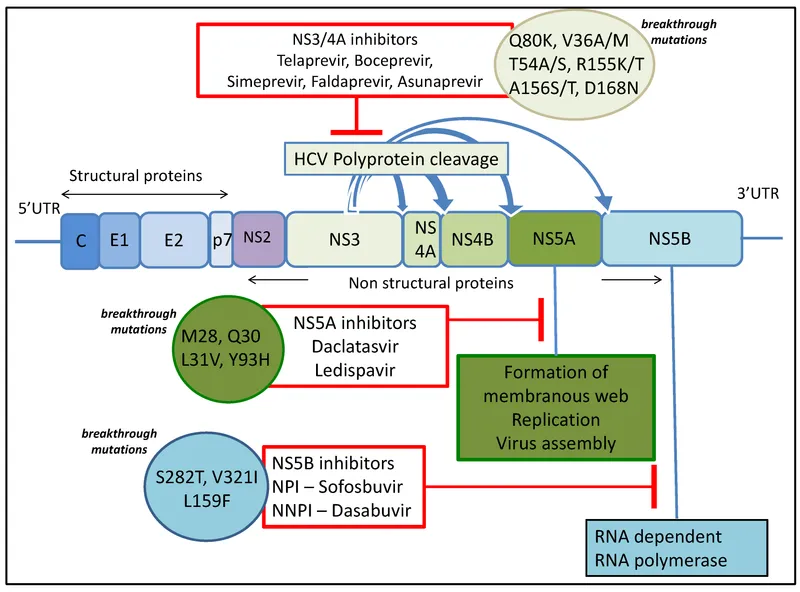
Direct-Acting Antivirals (DAAs) target specific HCV non-structural proteins to halt viral replication. Key classes include:
- NS3/4A Protease Inhibitors (-previr): E.g., Glecaprevir. Block post-translational processing of the HCV polyprotein.
- NS5A Inhibitors (-asvir): E.g., Pibrentasvir. Interfere with viral replication and assembly.
- NS5B Polymerase Inhibitors (-buvir): E.g., Sofosbuvir. Inhibit the RNA-dependent RNA polymerase, terminating chain elongation.
📌 Previr = Protease | Asvir = NS5A | Buvir = NS5B.
⭐ Sofosbuvir, a nucleotide analog NS5B inhibitor, has a high genetic barrier to resistance, making it a cornerstone of many DAA regimens.
Mechanisms of Resistance - Viral Escape Tactics
- Genetic Plasticity: HCV's high replication rate (≈$10^{12}$ virions/day) and error-prone RNA polymerase (NS5B) create a diverse viral "quasispecies," ripe for mutations.
- Resistance-Associated Variants (RAVs): These are mutations in DAA target regions (NS3/4A, NS5A, NS5B) that reduce drug susceptibility.
- Pre-existing: Naturally present at low levels before therapy begins.
- Emergent: Selected for and become dominant under the pressure of DAA treatment, leading to virologic failure.
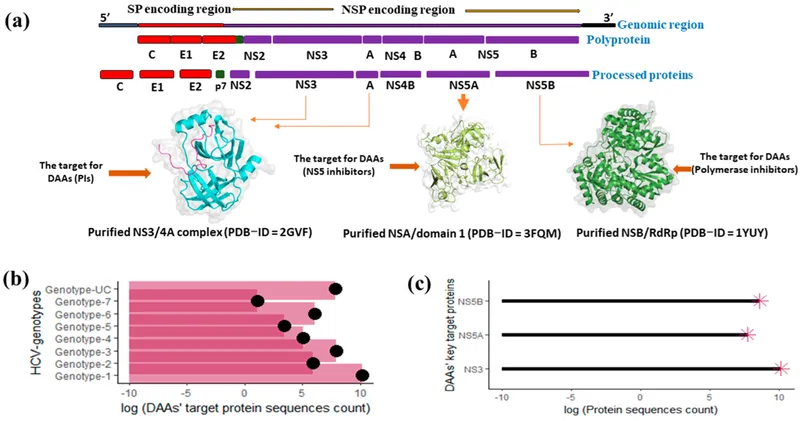
⭐ NS5A RAVs: Mutations in the NS5A region (e.g., Y93H) are a major cause of treatment failure for many DAA regimens. They can confer high-level resistance and persist for years after treatment cessation.
Clinically Significant RAVs - The Usual Suspects
-
NS5A Inhibitors (-asvirs): Low Barrier to Resistance
- Most common site for clinically significant RAVs. Single amino acid changes can confer high-level resistance.
- Key RAVs include Q30R, L31M/V, and Y93H/N.
- These variants primarily impact the "-asvir" class (e.g., Ledipasvir, Daclatasvir).
-
NS5B Polymerase Inhibitors (-buvirs): High Barrier to Resistance
- Sofosbuvir is robust against resistance; clinically significant RAVs are rare.
- The key (but unfit and rare) mutation is S282T.
⭐ Pre-treatment RAV testing (especially for NS5A variants) is crucial in specific scenarios, such as prior DAA failure or when considering certain regimens like Elbasvir/Grazoprevir for HCV genotype 1a.
Managing Treatment Failure - Salvage & Strategy
- Post-Failure Workup: Test for Resistance-Associated Variants (RAVs), especially in the NS5A gene, to guide subsequent therapy selection.
- Salvage Principles:
- Switch to different DAA classes not previously used.
- Use potent, pangenotypic regimens.
- Consider extending treatment duration (e.g., 12-16 weeks) or adding Ribavirin (RBV), especially with cirrhosis or complex RAVs.
⭐ Sofosbuvir/velpatasvir/voxilaprevir (Vosevi) is a highly effective single-tablet salvage regimen for patients who have failed prior DAA therapy, including those with NS5A RAVs.
High‑Yield Points - ⚡ Biggest Takeaways
- HCV's high mutation rate, via its error-prone RNA polymerase (RdRp), is the primary driver of resistance.
- Resistance-Associated Substitutions (RASs) can emerge during therapy, leading to treatment failure.
- NS5A inhibitor resistance is common and clinically significant, sometimes requiring baseline RAS testing.
- Mutations in NS5B polymerase and NS3/4A protease also confer resistance to their respective DAAs.
- Treatment failure requires switching to a DAA regimen with a different mechanism of action.
- Salvage therapy may involve combining DAAs, often with ribavirin.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

