Heritability & Risk - It Runs in the Family
- Schizophrenia has a strong genetic component, demonstrated by family, twin, and adoption studies. It follows a polygenic inheritance model, not a simple Mendelian pattern.
- Lifetime risk is directly proportional to the degree of genetic relatedness:
- General Population: 1%
- Sibling with schizophrenia: 9%
- Child with one parent with schizophrenia: 13%
- Dizygotic twin: ~17%
- Monozygotic twin: ~48-50%
⭐ The fact that monozygotic twin concordance is only ~50%, not 100%, is a classic exam point. It strongly implicates non-genetic factors (e.g., environment, epigenetics) in the disease's etiology.
Gene Hotspots - Chromosomal Culprits
- Highly polygenic, with heritability estimated at ~80%. No single gene is causative.
- Key chromosomal microdeletions confer the highest risk:
- 22q11.2 Deletion Syndrome: The single strongest genetic predictor. Also known as Velocardiofacial or DiGeorge syndrome.
- 16p11.2 Duplication/Deletion
- 1q21.1 & 3q29 Deletions
- Specific gene associations:
- MHC region on Chromosome 6: Genes like C4 are implicated in excessive synaptic pruning.
- DISC1 (Disrupted in Schizophrenia 1)
- NRG1 (Neuregulin 1)
⭐ The risk of schizophrenia in individuals with 22q11.2 deletion is 25-30%, a nearly 30-fold increase over the general population's 1% risk.
Syndromic Links - When Genes Go Wrong
- 22q11.2 Deletion Syndrome (VCFS/DiGeorge): The most significant known syndromic cause linked to schizophrenia.
- Individuals have a ~25-30% lifetime risk of developing schizophrenia.
- 📌 CATCH-22 Mnemonic: Cardiac defects, Abnormal facies, Thymic hypoplasia, Cleft palate, Hypocalcemia.
- Other Associated Syndromic Risks:
- Fragile X Syndrome: FMR1 gene premutation carriers show ↑ rates of psychosis.
- Klinefelter Syndrome (XXY): Associated with a 4-fold ↑ risk of psychotic disorders.
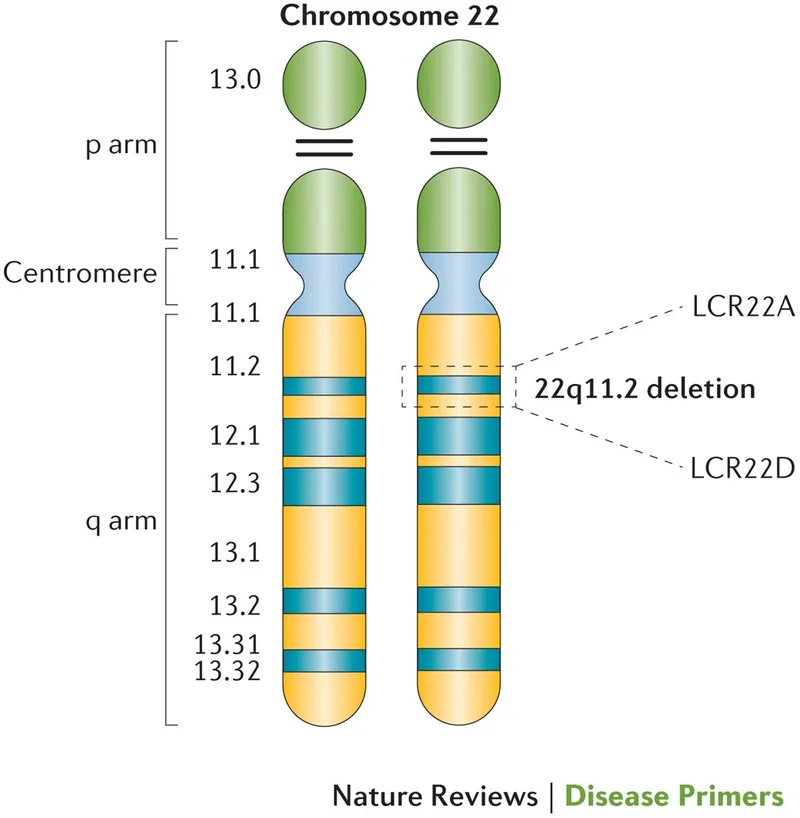
⭐ High-Yield: Psychosis in 22q11.2 deletion syndrome often presents earlier than idiopathic schizophrenia and may have more prominent negative and cognitive symptoms.
Epigenetics & Models - Nature Meets Nurture
- Gene-Environment Interaction: Genetic predisposition isn't destiny. Environmental factors modulate gene expression via epigenetic changes.
- Key Mechanisms:
- DNA Methylation: ↑ methylation can silence protective genes.
- Histone Modification: Acetylation/deacetylation alters chromatin, affecting gene access.
- Environmental Triggers:
- Prenatal: Maternal infection (influenza), malnutrition.
- Adolescence: Cannabis use, urban upbringing, social stress.
- "Two-Hit" Hypothesis:
- First Hit: Genetic or developmental disruption (prenatal).
- Second Hit: Environmental stressor (e.g., adolescent drug use) triggers psychosis onset.
⭐ Advanced paternal age (>50) is linked to a higher risk of de novo mutations in sperm, contributing to schizophrenia risk in offspring.
- Schizophrenia has a strong genetic component with heritability estimated at ~80%.
- Monozygotic twin concordance is ~40-50%, while dizygotic twin concordance is ~10-15%.
- Having a first-degree relative with schizophrenia is the single strongest risk factor for developing the illness.
- The disorder is polygenic; it is not caused by a single gene but multiple genes of small effect.
- Specific genetic syndromes like 22q11.2 deletion are associated with an increased risk.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

