Acid-Base Basics - The pH Party
- Acid: A proton ($H⁺$) donor.
- Base: A proton ($H⁺$) acceptor.
- pH: The measure of acidity, defined as $pH = -log[H⁺]$.
- Normal arterial blood pH: 7.35-7.45.
- Buffer System: A solution that resists pH change by neutralizing added acid or base. The primary buffer in the ECF is the bicarbonate system ($H₂CO₃/HCO₃⁻$).
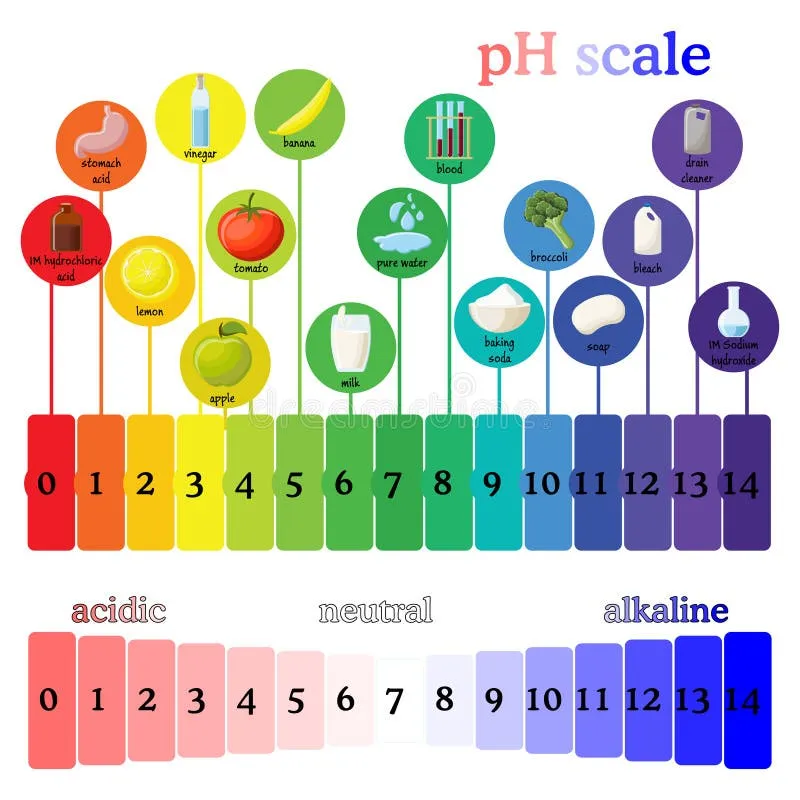
⭐ A pH change of 0.3 units corresponds to a 2x change in $[H⁺]$.
The Main Event - H-H Equation
-
The Equation: A fundamental formula used to calculate the pH of a buffer solution, linking pH to the ratio of the buffer system's components.
- General Form: $pH = pKa + log([A⁻]/[HA])$
- Bicarbonate System: $pH = 6.1 + log([HCO₃⁻]/(0.03 \times PCO₂))$
-
Components Defined:
- pKa: Acid dissociation constant; 6.1 for the bicarbonate system.
- [A⁻]: Conjugate base (bicarbonate, HCO₃⁻).
- [HA]: Weak acid (carbonic acid, proportional to PCO₂).
-
Primary Use: Crucial for calculating the pH of blood and understanding how the bicarbonate buffer system counteracts changes in acidity.
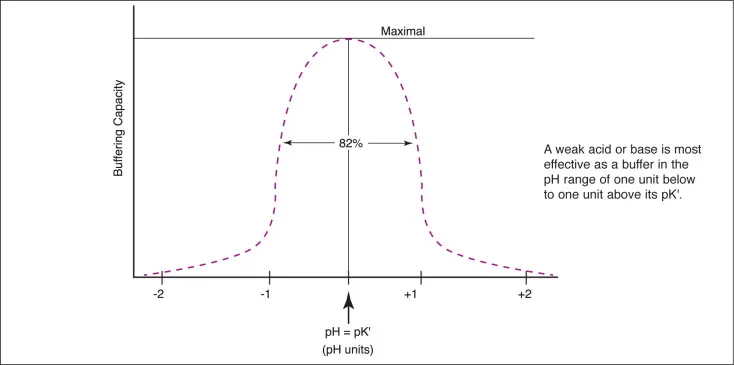
⭐ High-Yield: The body maintains a [HCO₃⁻] to (0.03 \times PCO₂) ratio of approximately 20:1 to keep blood pH around 7.4. When [A⁻] = [HA], pH = pKa, which represents the point of maximum buffering capacity.
Body's Buffer - Bicarb Brigade
-
The most crucial extracellular buffer for maintaining blood pH within the narrow range of 7.35-7.45.
-
Henderson-Hasselbalch Equation (Physiological):
- $pH = 6.1 + log([HCO₃⁻]/(0.03 * PCO₂))$
- This equation links pH to the ratio of the metabolic base ($[HCO₃⁻]$) and the respiratory acid ($PCO₂$).
-
Organ Regulation:
- Lungs (Fast): Regulate $PCO₂$ via ventilation. Changes occur in minutes.
- Kidneys (Slow): Regulate $[HCO₃⁻]$ by reabsorbing or excreting it. Takes hours to days.
- 📌 Mnemonic: ROME
- Respiratory Opposite: pH and $PCO₂$ move in opposite directions.
- Metabolic Equal: pH and $[HCO₃⁻]$ move in the same direction.
⭐ The pKa of the bicarbonate system is 6.1, far from physiological pH (7.4). Its power lies in the independent and tight regulation of both $PCO₂$ (lungs) and $[HCO₃⁻]$ (kidneys).
Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| Disorder | Primary Change | pH Trend | Compensation |
|---|---|---|---|
| Metabolic Acidosis | ↓ $[HCO₃⁻]## Clinical Correlation - pH-inding Problems |
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| ↓ | ↓ $PCO₂## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| | Metabolic Alkalosis | ↑ $[HCO₃⁻]## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| ↑ | ↑ $PCO₂## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| | Respiratory Acidosis | ↑ $PCO₂## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| ↓ | ↑ $[HCO₃⁻]## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| | Respiratory Alkalosis | ↓ $PCO₂## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
| ↑ | ↓ $[HCO₃⁻]## Clinical Correlation - pH-inding Problems
-
The Henderson-Hasselbalch equation is the key to interpreting arterial blood gas (ABG) results. Normal values are critical:
- pH: 7.35-7.45
- $PCO₂$: 35-45 mmHg
- $[HCO₃⁻]$: 22-26 mEq/L
-
Primary acid-base disorders are defined by the initial change in $PCO₂$ (respiratory) or $[HCO₃⁻]$ (metabolic).
|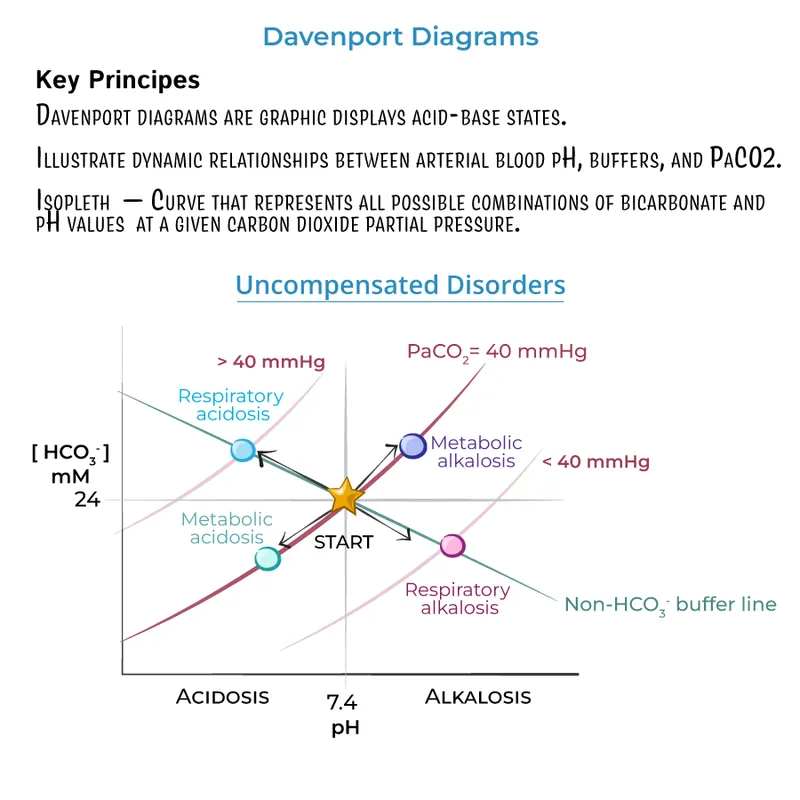
⭐ In metabolic acidosis, always calculate the anion gap ($Na⁺ - (Cl⁻ + HCO₃⁻)$). A high anion gap (HAGMA) indicates the addition of an unmeasured acid (e.g., MUDPILES).
High‑Yield Points - ⚡ Biggest Takeaways
- The Henderson-Hasselbalch equation links pH, pKa, and the bicarbonate buffer system.
- It calculates blood pH using the ratio of bicarbonate [HCO₃⁻] (metabolic component) to dissolved CO₂ (respiratory component).
- The key is the ratio of base to acid, not their absolute values.
- A 20:1 ratio of [HCO₃⁻] to dissolved PCO₂ maintains a normal blood pH of 7.4.
- Metabolic acidosis/alkalosis primarily alters [HCO₃⁻].
- Respiratory acidosis/alkalosis primarily alters PCO₂.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

