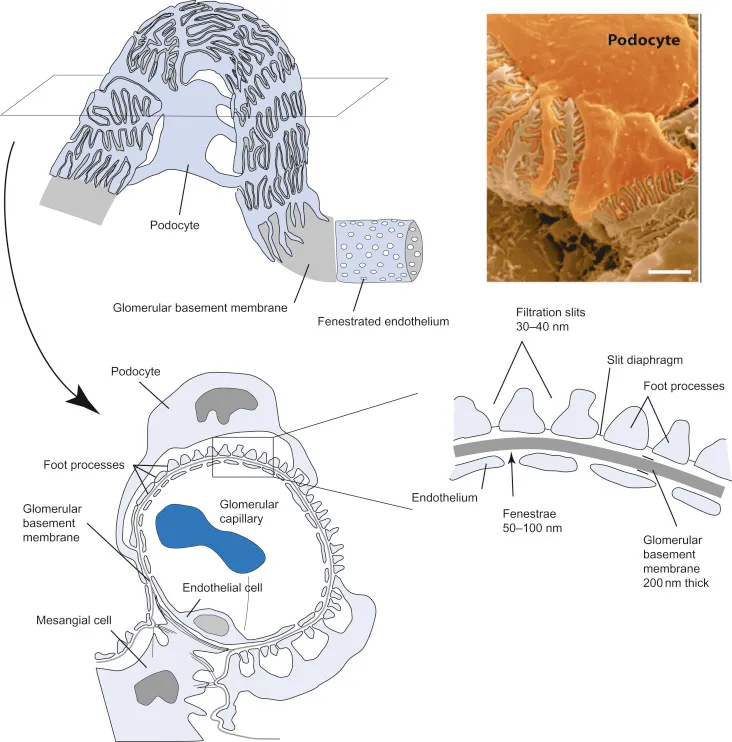
Glomerular Filtration - The Kidney's Sieve
- The initial, passive step of renal excretion where blood plasma and small molecules are forced from the glomerulus into Bowman's capsule.
- Filtration Barrier Selectivity: Based on size and charge.
- Freely filtered: Unbound drugs with molecular weight < 60 kDa.
- Not filtered: Drugs bound to albumin, large molecules (e.g., heparin), and blood cells.
- The rate is determined by the Glomerular Filtration Rate (GFR), driven by hydrostatic pressure.
- Filtration Fraction represents the proportion of renal plasma flow that is filtered: $FF = GFR / RPF$.
⭐ The glomerular basement membrane (GBM) is negatively charged, which electrostatically repels other anions like albumin, preventing their filtration even if they are small enough to pass through the pores.
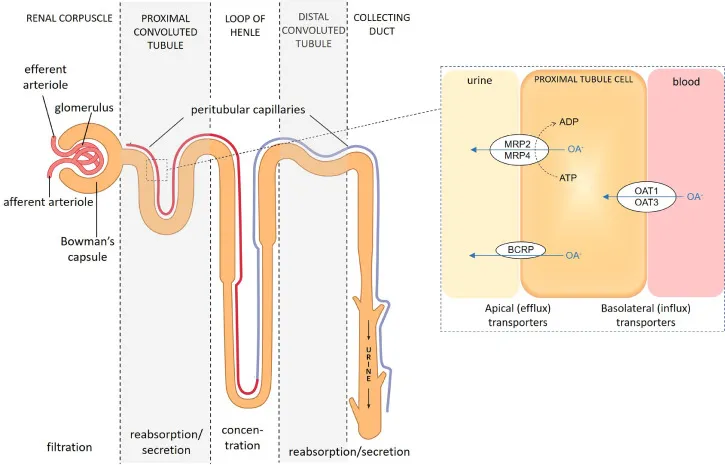
Proximal Tubular Secretion - Active Drug Dumping
- An active transport process in the proximal convoluted tubule (PCT) that "pumps" drugs from blood into urine, often against a concentration gradient.
- It's energy-dependent (ATP), carrier-mediated, and can be saturated at high drug concentrations.
- Low substrate specificity allows for rapid elimination of many xenobiotics.
- Two main transporter families:
- Organic Anion Transporters (OATs): Secrete weak acids like Penicillin, Furosemide, Salicylates, and Methotrexate.
- Organic Cation Transporters (OCTs): Secrete weak bases like Metformin, Morphine, Amiloride, and Procainamide.
⭐ Competition for OATs is a classic drug-drug interaction source. Probenecid blocks Penicillin secretion, ↑ its half-life and efficacy. It also blocks uric acid reabsorption, making it a key gout treatment.
Tubular Reabsorption - The pH Power Play
- Principle: Lipid-soluble (non-ionized) drugs passively diffuse from the tubular lumen back into the blood. Water-soluble (ionized) drugs are "trapped" in the tubule and excreted.
- Urine pH determines a drug's ionization state. We can manipulate urine pH to accelerate the excretion of specific drugs during overdose, a principle known as ion trapping.
- Weak Acids (e.g., Aspirin, phenobarbital):
- To ↑ excretion, alkalinize the urine (e.g., sodium bicarbonate).
- In alkaline urine, weak acids become ionized ($A^-$) and are trapped.
- $HA \rightleftharpoons H^+ + A^-$
- Weak Bases (e.g., Amphetamines, TCAs, PCP):
- To ↑ excretion, acidify the urine (e.g., ammonium chloride, vitamin C).
- In acidic urine, weak bases become ionized ($BH^+$) and are trapped.
- $B + H^+ \rightleftharpoons BH^+$
⭐ In aspirin (salicylate) overdose, IV sodium bicarbonate is critical. It alkalinizes the urine, trapping the ionized aspirin in the tubule for rapid excretion.
📌 To trap a drug, make the urine the opposite pH.
Clearance Calculation - Excretion by Numbers
- Clearance (CL): Volume of plasma cleared of drug per unit time (mL/min).
- Formula: $CL = (\text{Rate of elimination}) / C$
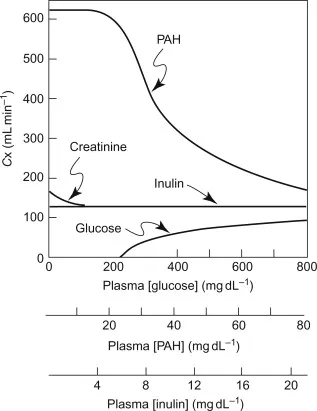
- Renal Handling: Compare drug clearance ($CL_x$) to GFR ($CL_{inulin}$ ≈ 125 mL/min).
- $CL_x < GFR$: Net tubular reabsorption.
- $CL_x = GFR$: Filtration only.
- $CL_x > GFR$: Net tubular secretion.
⭐ Para-aminohippuric acid (PAH) clearance approximates renal plasma flow (RPF) as it is both filtered and secreted (~650 mL/min).
High‑Yield Points - ⚡ Biggest Takeaways
- Glomerular filtration is a passive process; only small, unbound drugs are filtered.
- Proximal tubular secretion is an active process using OATs (for weak acids) and OCTs (for weak bases).
- Distal tubular reabsorption is passive for lipid-soluble, non-ionized drugs.
- Alkalinize urine (e.g., NaHCO₃) to ↑ excretion of weak acids (aspirin) via ion trapping.
- Acidify urine (e.g., NH₄Cl) to ↑ excretion of weak bases (amphetamines) via ion trapping.
- Renal disease ↓ drug clearance, often requiring dose reduction.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

