Phase I Intro - Making Drugs Polar
- Goal: ↑ drug polarity & water solubility to facilitate excretion.
- Mechanism: Introduces or unmasks polar functional groups (e.g., -OH, -NH₂, -SH).
- Key Reactions: 📌 ROH
- Reduction
- Oxidation (most common, uses CYP450)
- Hydrolysis
- The resulting metabolite can be active, inactive, or toxic. It's now prepped for Phase II conjugation or direct renal elimination.
⭐ Most Phase I reactions are catalyzed by the Cytochrome P450 (CYP450) enzyme system located in the smooth endoplasmic reticulum of hepatocytes.
Cytochrome P450 - The Liver's MVP
- Heme-containing monooxygenase superfamily in the hepatic smooth endoplasmic reticulum.
- Catalyzes the oxidation of many drugs, facilitating their excretion.
- General Reaction: $Drug + O_2 + NADPH + H^+ \rightarrow Drug-OH + H_2O + NADP^+$
- Inducers (↑ metabolism): Rifampin, Carbamazepine, Phenobarbital, Phenytoin, St. John's Wort.
- Inhibitors (↓ metabolism): Grapefruit juice, Cimetidine, Macrolides, Azole antifungals, Protease inhibitors.
- 📌 Mnemonic (Inhibitors): CRACK AMIGOS
⭐ Exam Favorite: CYP3A4 is the most abundant isoform, metabolizing over 50% of clinically used drugs. Its inhibition (e.g., by grapefruit juice) can lead to toxic drug levels.
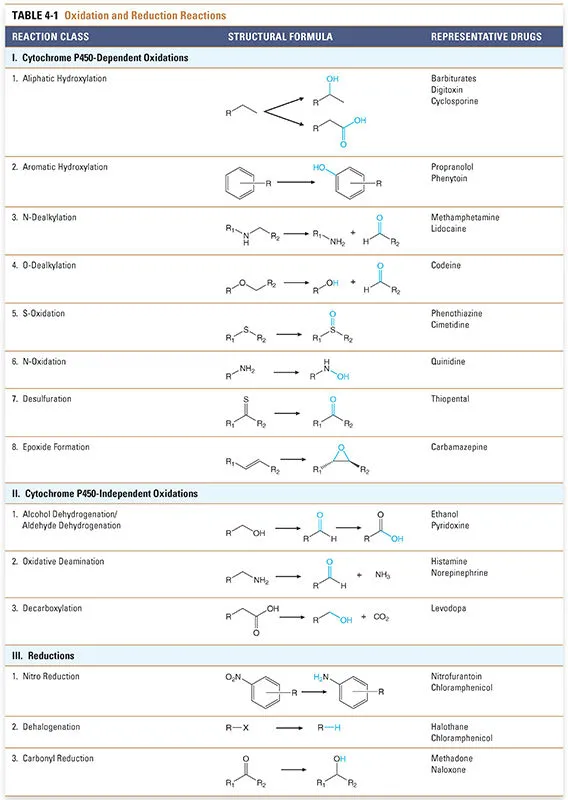
The Reactions - Oxidation, Reduction, Hydrolysis
- Oxidation: The most common reaction, primarily via Cytochrome P450 (CYP450) enzymes.
- Adds or unmasks polar functional groups (e.g., -OH, -NH2).
- Reactions include hydroxylation, dealkylation, and deamination.
- Reduction: Less frequent; involves adding hydrogen or removing oxygen.
- Catalyzed by reductases (e.g., carbonyl reductase).
- Example: Naloxone metabolism.
- Hydrolysis: Cleavage by adding water, crucial for esters and amides.
- Occurs in the liver, plasma, and GI tract.
- Mediated by esterases (e.g., for aspirin) and amidases (e.g., for procainamide).
⭐ Phase I reactions don't always inactivate a drug; they can convert a prodrug into its active form (e.g., codeine to morphine).
Clinical Impact - Inducers & Inhibitors
| Feature | Enzyme Inducers | Enzyme Inhibitors |
|---|---|---|
| Mechanism | ↑ Synthesis of CYP450 enzymes | ↓ Activity of CYP450 enzymes |
| Effect | ↑ Drug metabolism → ↓ drug levels | ↓ Drug metabolism → ↑ drug levels |
| Result | Sub-therapeutic effect or "tolerance" | Increased risk of toxicity |
| Onset | Slow (days to weeks) | Fast (hours to days) |
| Mnemonic 📌 | CRAP GPS: Chronic alcohol, Rifampin, Anticonvulsants (Carbamazepine, Phenobarbital), Griseofulvin, Phenytoin, St. John's Wort | SICKFACES.COM + G: Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Acute alcohol, Chloramphenicol, Erythromycin, Sulfonamides, .COM (Ciprofloxacin, Omeprazole, Metronidazole), + Grapefruit juice |
High‑Yield Points - ⚡ Biggest Takeaways
- Phase I reactions aim to increase polarity of drugs by unmasking or adding functional groups (-OH, -NH₂).
- The cytochrome P450 (CYP450) system, located in the hepatic smooth ER, is the primary catalyst for these reactions.
- Oxidation is the most common Phase I reaction; reduction and hydrolysis are others.
- These reactions can activate prodrugs or prepare compounds for Phase II conjugation.
- CYP inhibitors (e.g., grapefruit juice, macrolides) can ↑ drug toxicity.
- CYP inducers (e.g., rifampin, St. John's wort) can cause ↓ therapeutic failure.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

