AUC Fundamentals - Total Drug Exposure
- Represents the total systemic exposure to a drug over a period of time; it reflects the extent of drug absorption.
- Calculated as the integral of the plasma drug concentration-time curve, often estimated using the trapezoidal rule.
- Directly proportional to the dose and inversely proportional to clearance ($CL$) for drugs with linear kinetics.
- $AUC = \frac{Dose}{CL}$
- Essential for bioavailability (F) studies, comparing drug formulations.
- $F = (\frac{AUC_{oral}}{AUC_{IV}}) \times 100%$
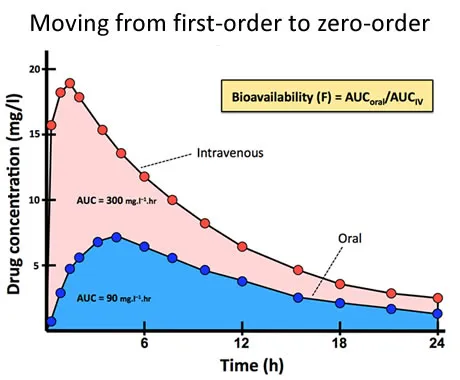
⭐ In steady-state, the AUC during a dosing interval ($AUC_{ss}$) is equal to the AUC from time zero to infinity after a single dose ($AUC_{0-\infty}$). This principle is key for designing multiple-dosing regimens.
AUC Calculation - The Trapezoid Rule
- Estimates Area Under the Curve (AUC) from a series of discrete plasma drug concentration ($C$) vs. time ($t$) data points.
- The method divides the concentration-time curve into several trapezoids and sums their individual areas to find the total.
- Single Trapezoid Area: $AUC_{t1 o t2} = \frac{(C_1 + C_2)}{2} \times (t_2 - t_1)$
- Extrapolation to Infinity: The final area segment, from the last measured point ($C_{last}$) to infinity, is calculated as $AUC_{t_{last} \to \infty} = \frac{C_{last}}{k_{el}}$.
- $k_{el}$ is the terminal elimination rate constant.
⭐ AUC is the most reliable measure of a drug's total systemic exposure over time. It is directly proportional to the dose and inversely proportional to the drug's clearance (CL).
- This relationship is fundamental for calculating bioavailability (F).
AUC & PK Parameters - Clearance & Bioavailability
-
Area Under the Curve (AUC): Represents total systemic drug exposure over time. It is directly proportional to the dose and bioavailability ($F$), and inversely proportional to clearance ($CL$).
- Used to calculate key parameters like bioavailability and clearance.
-
Clearance (CL): The theoretical volume of plasma cleared of a drug per unit time (e.g., L/hr).
- Formula: $CL = (Dose \times F) / AUC$
- For IV administration, bioavailability ($F$) is 100% (or 1), so the formula simplifies to $CL = Dose_{IV} / AUC_{IV}$.
-
Bioavailability (F): The fraction of an administered dose reaching systemic circulation.
- Formula: $F = \frac{AUC_{oral} \times Dose_{IV}}{AUC_{IV} \times Dose_{oral}}$
⭐ Drugs with high first-pass metabolism (e.g., nitroglycerin, lidocaine) have low oral bioavailability ($F$). This leads to a much smaller $AUC_{oral}$ compared to $AUC_{IV}$ for an equivalent dose, often necessitating non-oral routes.
Clinical Use - Bioequivalence & TDM
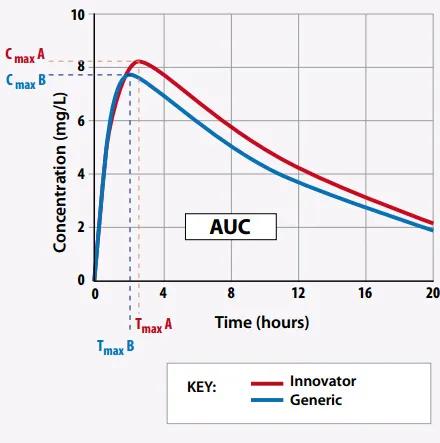
- Bioequivalence: Assessed when comparing generic to brand-name drugs.
- Requires similar AUC, Cmax, and Tmax.
- FDA accepts if the 90% CI of the ratio for AUC and Cmax falls within 80-125%.
- Therapeutic Drug Monitoring (TDM):
- Guides dosing for drugs with a narrow therapeutic index (NTI) to maintain efficacy and avoid toxicity.
- 📌 NTI Drugs: Warfarin, Theophylline, Digoxin, Phenytoin, Lithium.
⭐ For IV administration, bioavailability (F) is 100%, and the formula simplifies to $AUC = Dose / CL$. This direct relationship is crucial for dose adjustments in TDM.
High‑Yield Points - ⚡ Biggest Takeaways
- Area Under the Curve (AUC) represents the total systemic drug exposure over time, integrating concentration and time.
- Bioavailability (F) is calculated by comparing the AUC of an extravascular route (e.g., oral) to the AUC of IV administration.
- Clearance (CL) is inversely related to AUC for a given dose; the core formula is CL = Dose / AUC.
- In multiple-dosing regimens, the AUC over a dosing interval at steady state is crucial for therapeutic monitoring.
- The trapezoidal rule is the most common method to estimate AUC from a concentration-time graph.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

