Immunosuppression Phases - The Game Plan
- Induction (Peri-operative): High-potency, short-term lymphocyte depletion to prevent hyperacute/acute rejection. Agents: Basiliximab (IL-2R Ab), Antithymocyte Globulin (ATG).
- Maintenance (Lifelong): Lower-dose combination therapy to prevent chronic rejection and minimize toxicity.
- Rejection Treatment (Episodic): High-dose pulse therapy for breakthrough rejection (e.g., steroids, ATG).
⭐ Most maintenance regimens use triple therapy: a Calcineurin Inhibitor (Tacrolimus), an Antiproliferative agent (Mycophenolate), and Corticosteroids.
Induction Therapy - The Shock Troops
- Goal: Provide intense, short-term immunosuppression immediately post-transplant. Aims to prevent acute rejection by depleting lymphocytes or blocking their activation.
- Key Agents:
- Biologics (Antibodies): Cornerstone of induction.
- IL-2 Receptor Antagonist: Basiliximab. Specifically targets the CD25 receptor on activated T-cells, preventing their proliferation.
- Polyclonal Antibodies: Antithymocyte Globulin (ATG) (rabbit/equine). Causes broad T-cell depletion.
- Anti-CD52 Antibody: Alemtuzumab. Depletes a wide range of lymphocytes.
- High-Dose Corticosteroids: e.g., Methylprednisolone, given intraoperatively and for a short period post-op.
- Biologics (Antibodies): Cornerstone of induction.
⭐ Basiliximab offers targeted therapy against activated T-cells only, sparing resting lymphocytes. This leads to fewer side effects like cytomegalovirus (CMV) infection compared to broadly depleting agents like ATG.
Maintenance Therapy - The Long Patrol
- Goal: Prevent acute & chronic rejection while minimizing long-term drug toxicity. This is a lifelong balancing act.
- Cornerstone Regimen (Triple Drug Therapy):
- Calcineurin Inhibitor (CNI): Tacrolimus (preferred) or Cyclosporine.
- Blocks IL-2 synthesis, preventing T-cell activation.
- ⚠️ Key toxicities: Nephrotoxicity, neurotoxicity, hypertension, hyperglycemia.
- Antiproliferative Agent: Mycophenolate Mofetil (MMF) or Azathioprine.
- Inhibits lymphocyte proliferation by blocking purine synthesis.
- ⚠️ Key toxicities: GI intolerance (diarrhea), bone marrow suppression.
- Corticosteroids: Prednisone.
- Broad anti-inflammatory effects; tapered to the lowest effective dose (e.g., ≤5 mg/day) or discontinued.
- Calcineurin Inhibitor (CNI): Tacrolimus (preferred) or Cyclosporine.
- CNI-Sparing Alternatives:
- mTOR inhibitors (Sirolimus, Everolimus) may replace CNIs to avoid renal toxicity, but have their own side effects (e.g., poor wound healing, hyperlipidemia).
⭐ Chronic allograft nephropathy is often multifactorial, but long-term CNI-induced nephrotoxicity is a major contributor and can be difficult to distinguish from chronic rejection.
Rejection Treatment - The Firefighters
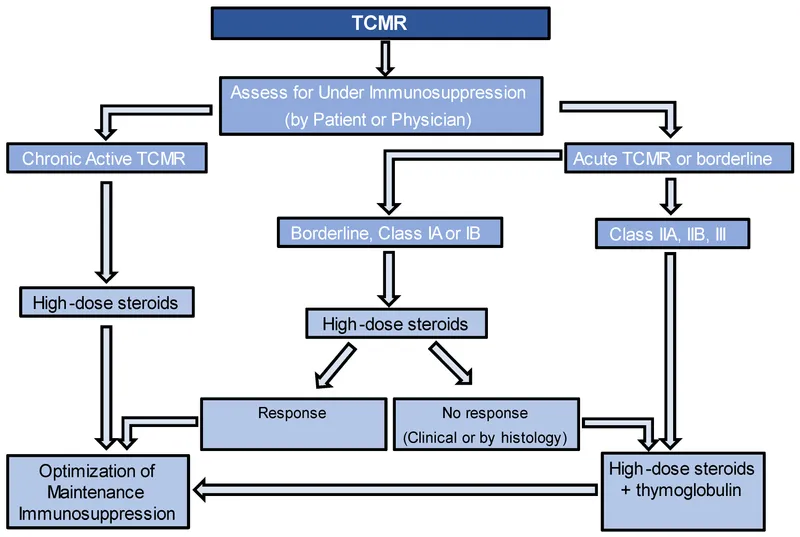
- Acute Cellular Rejection: Primarily T-cell mediated. Treated with high-dose corticosteroids.
- Antibody-Mediated (Humoral) Rejection: Involves B-cells and antibodies. Requires plasmapheresis, IVIG, and often rituximab.
⭐ Anti-thymocyte globulin (ATG) can induce a "cytokine release syndrome" (fever, chills, hypotension) on first infusion. Pre-medicate with corticosteroids, acetaminophen, and diphenhydramine.
High‑Yield Points - ⚡ Biggest Takeaways
- Standard triple therapy combines a calcineurin inhibitor (CNI), an antimetabolite, and corticosteroids.
- CNI-induced nephrotoxicity is the most significant long-term complication of tacrolimus and cyclosporine.
- Mycophenolate is a potent antimetabolite but is limited by GI intolerance and myelosuppression.
- Sirolimus (mTOR inhibitor) is used to spare renal function but can cause hyperlipidemia and impaired wound healing.
- Induction therapy (e.g., basiliximab) prevents acute rejection by targeting IL-2 receptors.
- Prophylaxis for PJP and CMV is essential in these patients.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

