Absorption Interactions - First-Pass Fumbles
-
Altered Gut Lumen Chemistry:
- Chelation: Drugs bind in the gut, preventing absorption.
- Example: Tetracyclines or fluoroquinolones + divalent cations (Ca²⁺, Mg²⁺, Fe²⁺) in antacids or dairy.
- Altered pH: Changed stomach acidity impairs absorption of pH-dependent drugs.
- Example: ↓ absorption of ketoconazole (needs acid) with proton pump inhibitors (PPIs).
- Chelation: Drugs bind in the gut, preventing absorption.
-
Altered Gut Motility:
- ↑ Motility: Less time for absorption (e.g., metoclopramide ↓ digoxin absorption).
- ↓ Motility: More time for absorption (e.g., anticholinergics ↑ digoxin absorption).
-
Gut Wall Metabolism (First-Pass Fumbles):
- Inhibition of enzymes in the gut wall (e.g., CYP3A4) increases bioavailability.
⭐ Grapefruit juice is a potent inhibitor of intestinal CYP3A4, significantly increasing levels of drugs like statins and calcium channel blockers, risking toxicity.
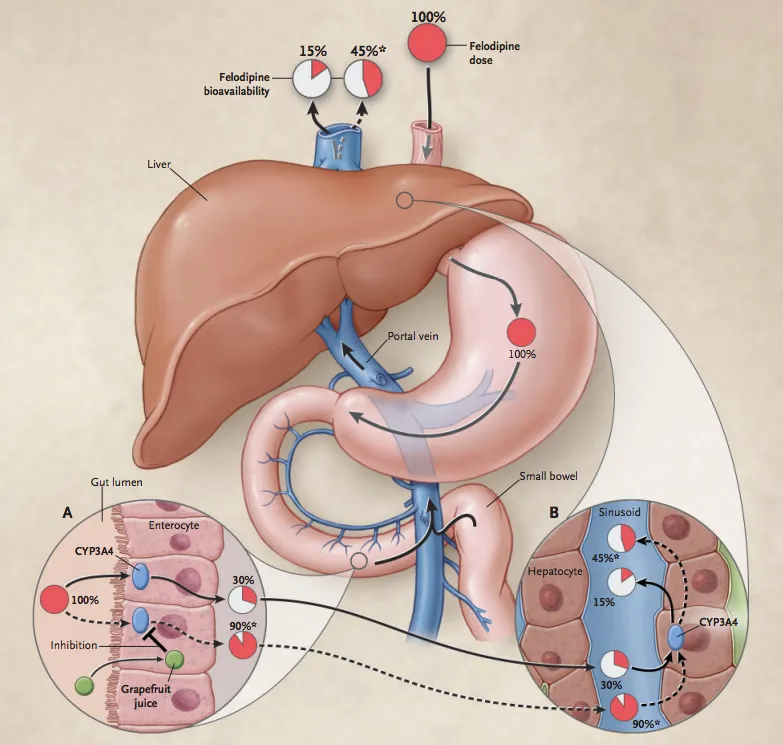
Distribution Interactions - Protein Binding Battles
- Mechanism: A highly protein-bound drug is displaced from plasma proteins (e.g., albumin) by another drug. This ↑ the free (active) concentration of the displaced drug.
- Equation: $Drug_{bound} ightleftharpoons Drug_{free} + Protein$
- High-Risk Drugs: Those with >90% protein binding and a narrow therapeutic index.
- Examples: Warfarin, Phenytoin, Methotrexate, Sulfonylureas.
- Classic Interaction: Sulfonamides or Aspirin displace Warfarin, leading to ↑ free Warfarin and ↑ bleeding risk.
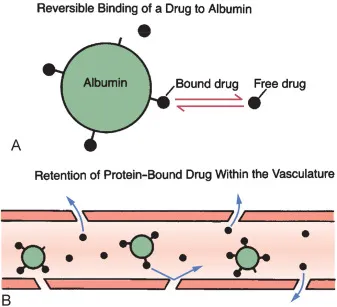
⭐ High-Yield Fact: The increase in free drug concentration is often transient. The newly freed drug is also more available for metabolism and excretion, which can lead to a new, lower total drug concentration steady-state over time.
Metabolism Interactions - CYP450 Power Plays
Cytochrome P450 (CYP450) enzymes are a major source of pharmacokinetic drug interactions. Modulating their activity can drastically alter drug concentrations, leading to toxicity or therapeutic failure.
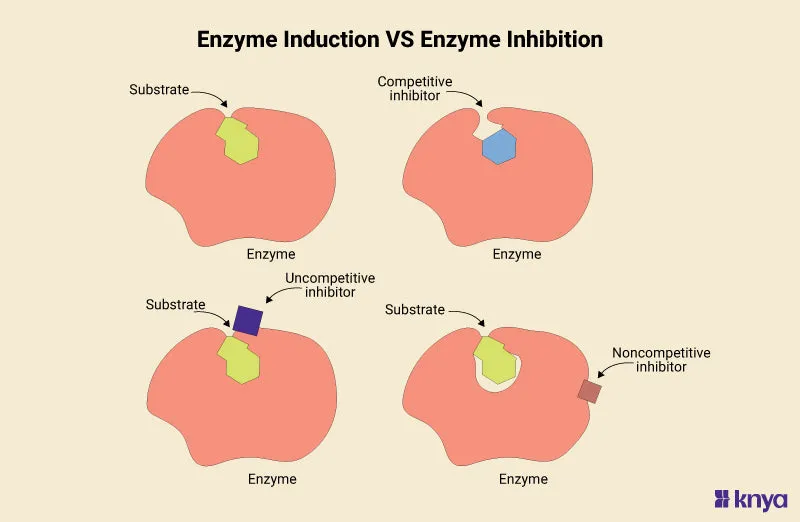
| CYP450 Inducers (↓ Drug Levels) | CYP450 Inhibitors (↑ Drug Levels) |
|---|---|
| Effect: Accelerate metabolism, ↓ substrate drug levels, potentially causing therapeutic failure. | Effect: Slow metabolism, ↑ substrate drug levels, potentially causing toxicity. |
| Examples: | Examples: |
| * Rifampin | * Macrolides (e.g., Erythromycin) |
| * Phenobarbital | * Azole antifungals (e.g., Ketoconazole) |
| * Carbamazepine | * Grapefruit juice |
| * St. John's Wort | * Protease Inhibitors (e.g., Ritonavir) |
| 📌 Mnemonic: CRAP GPS (Carbamazepine, Rifampin, Alcohol (chronic), Phenytoin, Griseofulvin, Phenobarbital, Sulfonylureas) | 📌 Mnemonic: SICKFACES.COM (Sodium valproate, Isoniazid, Cimetidine, Ketoconazole, Fluconazole, Alcohol (acute), Chloramphenicol, Erythromycin, Sulfonamides, Ciprofloxacin, Omeprazole, Metronidazole) |
Excretion Interactions - Kidney's Gatekeepers
-
Competition for Renal Transporters: Drugs can compete for active secretion into the renal tubules, primarily via Organic Anion Transporters (OATs) and Organic Cation Transporters (OCTs) in the proximal tubule.
- Example: Probenecid ↓ renal clearance of Penicillin by inhibiting OAT-mediated secretion, thus ↑ its plasma concentration and half-life.
-
Alteration of Urine pH (Ion Trapping): Modifying urine pH changes the ionization state of weak acids/bases, affecting their reabsorption.
- Weak Acids (e.g., Aspirin, Phenobarbital): Alkalinize urine with sodium bicarbonate ($HCO_3^−$) to ↑ excretion. $HA \rightleftharpoons H^+ + A^-$. Traps drug in its ionized, water-soluble form ($A^-$).
- Weak Bases (e.g., Amphetamines): Acidify urine (e.g., with ammonium chloride, $NH_4Cl$) to ↑ excretion.
⭐ For salicylate (aspirin) toxicity, administer sodium bicarbonate to alkalinize the urine, which promotes ion trapping and enhances renal excretion.
High‑Yield Points - ⚡ Biggest Takeaways
- CYP450 enzyme induction (e.g., rifampin) ↓ drug levels, while inhibition (e.g., grapefruit juice, azoles) ↑ toxicity.
- P-glycoprotein (P-gp) inhibition ↑ drug absorption and bioavailability; induction does the opposite.
- Altered absorption occurs via chelation (tetracyclines + iron) or gastric pH changes (antacids).
- Displacement from plasma proteins (e.g., warfarin) ↑ free drug concentration and toxicity risk.
- Altering urinary pH modifies the renal clearance of weak acids/bases (e.g., aspirin).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

