Mechanism of Action - The Viral Achilles' Heels
DAAs directly target key Hepatitis C non-structural (NS) proteins, halting viral replication.
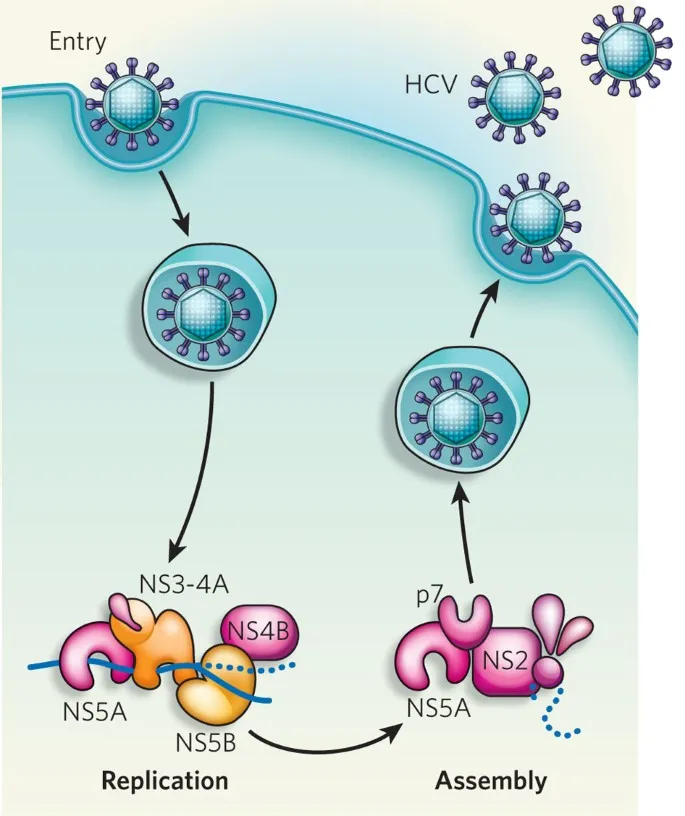
- NS3/4A Protease Inhibitors (-previr): Block post-translational processing of the HCV polyprotein. 📌 Previr for Protease.
- NS5A Inhibitors (-asvir): Disrupt the NS5A protein's role in viral RNA replication and virion assembly.
- NS5B Polymerase Inhibitors (-buvir): Inhibit the RNA-dependent RNA polymerase (RdRp), preventing synthesis of new HCV RNA.
⭐ DAAs are curative, achieving a sustained virologic response (SVR)-undetectable HCV RNA 12 weeks post-therapy-in >95% of cases. This is a virologic cure.
The 'Virs' - A Viral Name Game
Direct-acting antivirals (DAAs) for Hepatitis C are classified by their target and corresponding suffix. This naming convention helps identify the drug's mechanism of action.
| Class (Target) | Suffix | Example Drugs |
|---|---|---|
| NS3/4A Protease Inhibitor | -previr | Grazoprevir, Glecaprevir |
| NS5A Inhibitor | -asvir | Ledipasvir, Pibrentasvir |
| NS5B Polymerase Inhibitor | -buvir | Sofosbuvir, Dasabuvir |
⭐ Sofosbuvir is a nucleotide analog polymerase inhibitor that requires intracellular phosphorylation, while Dasabuvir is a non-nucleoside inhibitor that does not.
Treatment Regimens - The Combo Punch
- Goal: Eradicate HCV by achieving Sustained Virologic Response (SVR), defined as undetectable HCV RNA 12 weeks post-treatment. This is considered a cure.
- Strategy: Combination therapy is mandatory. Monotherapy is ineffective due to the rapid selection of drug-resistant viral variants.
- Pangenotypic Regimens: First-line standard of care, effective against all major HCV genotypes (1-6), simplifying the treatment approach.
- Glecaprevir + Pibrentasvir
- Sofosbuvir + Velpatasvir
⭐ Black Box Warning: All patients must be tested for current or prior Hepatitis B virus (HBV) infection before initiating DAAs. Treatment can cause HBV reactivation, leading to fulminant hepatitis and death.
Adverse Effects - The Payoff & Perils
-
Generally Well-Tolerated: Most adverse effects are mild and transient.
- Common complaints: Headache, fatigue, nausea.
- Less common: Rash, insomnia, diarrhea.
-
⚠️ Black Box Warning: HBV Reactivation
- DAAs can reactivate Hepatitis B virus in co-infected patients, potentially leading to fulminant hepatitis and liver failure.
- Action: All patients must be tested for current or prior HBV infection (HBsAg and anti-HBc) before starting therapy.
⭐ Drug Interactions: Co-administration of sofosbuvir with amiodarone is not recommended due to risk of severe symptomatic bradycardia.
- Direct-acting antivirals (DAAs) target HCV non-structural proteins: NS3/4A protease (-previr), NS5A (-asvir), and NS5B polymerase (-buvir).
- DAA therapy always uses a combination of agents from different classes to prevent resistance and achieve high efficacy.
- These regimens achieve >95% cure rates, defined as a Sustained Virologic Response (SVR).
- Black Box Warning: Risk of Hepatitis B reactivation; screen all patients for HBV before starting.
- Watch for significant drug-drug interactions, especially amiodarone with sofosbuvir (severe bradycardia) and CYP450 inducers/inhibitors.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

