IRI: The Basics - Comeback Kid's Curse
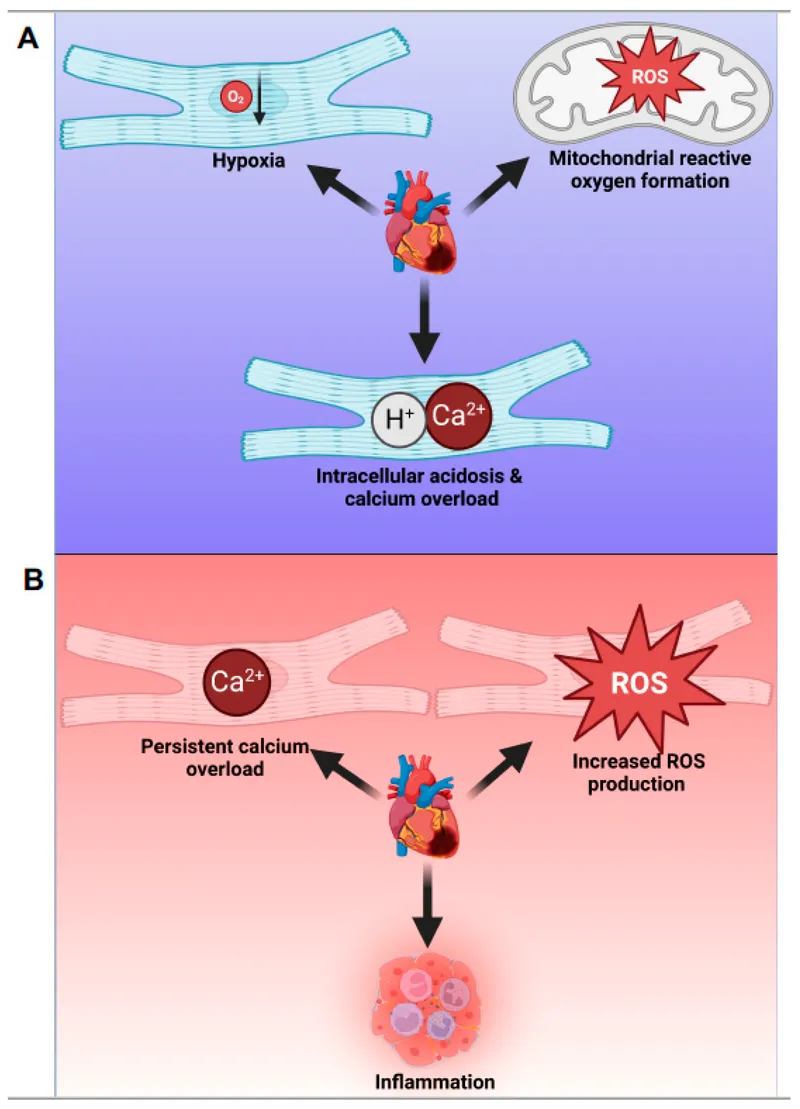
Paradoxical exacerbation of cell injury after restoring blood flow to ischemic tissue. The very act of reperfusion triggers a cascade of damage, driven by several key mechanisms.
- Core Mechanisms: 📌 RICM of Injury
- Reactive Oxygen Species (ROS): Sudden O₂ influx fuels a burst of ROS from mitochondria & neutrophils.
- Inflammation: Complement activation (C5a) and cytokines recruit neutrophils, which release proteases and more ROS.
- Ca²⁺ Overload: Ischemic pump failure leads to ↑ intracellular Ca²⁺; reperfusion worsens this, activating damaging enzymes.
- Mitochondrial Dysfunction: Ca²⁺ and ROS trigger the opening of the Mitochondrial Permeability Transition Pore (MPTP), leading to apoptosis.
⭐ Reperfusion can cause microvascular injury and endothelial swelling, leading to impaired blood flow in small vessels-the "no-reflow" phenomenon-despite restoration of circulation in larger arteries.
IRI: Damage Deep-Dive - The Oxygen Paradox
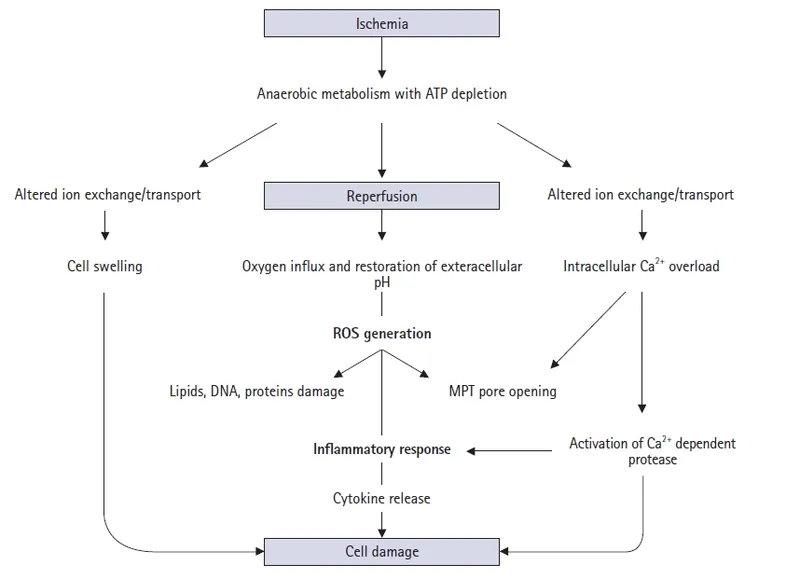
Re-introducing O₂ to ischemic tissues paradoxically worsens injury. Key drivers:
-
Reactive Oxygen Species (ROS) Burst:
- Incomplete reduction of O₂ by damaged mitochondria generates superoxide ($O_2^•−$).
- Inflammatory cells (neutrophils) produce ROS via NADPH oxidase.
- Leads to lipid peroxidation → membrane damage.
-
Inflammation Amplification:
- Reperfusion recruits neutrophils.
- Release of proteases & elastases damages endothelium and parenchyma.
- Cytokine release (TNF, Interleukins) fuels the inflammatory fire.
-
Complement Activation:
- Ischemia promotes deposition of IgM antibodies on endothelial cells.
- Reperfusion activates complement pathway → C5a (chemoattractant) & MAC (cell lysis).
⭐ High-Yield: A major source of damage in IRI is the respiratory burst from recruited neutrophils, which use the enzyme NADPH oxidase to generate a flood of superoxide radicals upon reperfusion.
IRI: Clinical Picture - Tissues Tell The Tale
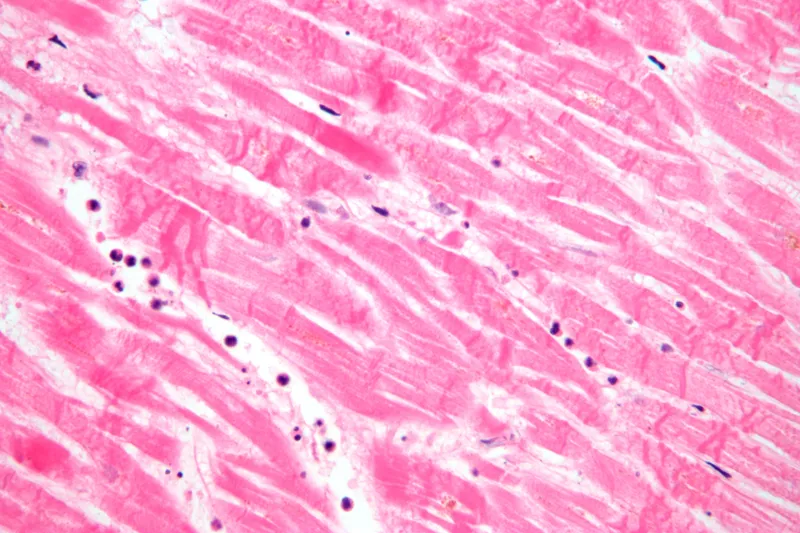
- Heart: Post-angioplasty, watch for reperfusion arrhythmias, myocardial stunning (prolonged dysfunction despite restored flow), and microvascular injury. Key histologic finding: contraction band necrosis.
- Brain: Following thrombolysis for stroke, can lead to hemorrhagic transformation and cerebral edema.
- Kidney: Ischemic acute tubular necrosis (ATN) can worsen despite re-established perfusion.
- Lungs & Gut: Reperfusion can trigger acute respiratory distress syndrome (ARDS) or increase gut permeability, risking sepsis.
⭐ Contraction band necrosis is a classic histologic sign of cardiac IRI. Massive Ca²⁺ influx upon reperfusion causes myocyte hypercontraction, creating distinct eosinophilic bands.
High‑Yield Points - ⚡ Biggest Takeaways
- Ischemia-reperfusion injury is a paradoxical increase in cell damage after restoring blood flow.
- Primarily driven by a surge in reactive oxygen species (ROS) from reperfused tissues and leukocytes.
- Mitochondrial permeability transition is a critical event, causing mitochondrial dysfunction and cell death.
- Features an intense inflammatory response, with neutrophil infiltration worsening the damage.
- Complement activation contributes to inflammation and direct cell injury.
- Clinically key in thrombolysis, angioplasty, and organ transplantation.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

