Indications for Invasion - Why We Test
- Advanced Maternal Age (AMA): Age ≥ 35 at delivery.
- Abnormal non-invasive screening test result (e.g., cfDNA, quad screen).
- Previous pregnancy with a documented chromosomal abnormality.
- Abnormal ultrasound findings:
- Structural defects (e.g., cardiac, neural tube).
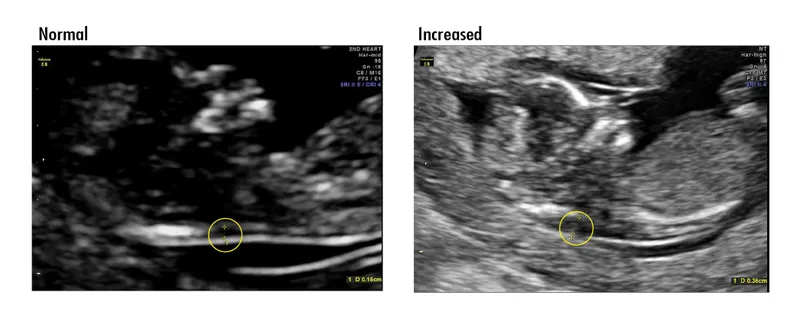
- Soft markers (e.g., ↑ nuchal translucency, absent nasal bone).
- Parental carrier of a chromosomal abnormality (e.g., balanced translocation).
📌 Mnemonic: AMAAP
⭐ The most common indication for invasive testing is now an abnormal cell-free DNA (cfDNA) result, surpassing AMA in many centers.
Chorionic Villus Sampling - The Early Peek
- Procedure: Transcervical or transabdominal aspiration of chorionic villi (placental tissue) for genetic/chromosomal analysis.
- Timing: Performed early at 10-13 weeks gestation.
- Advantages: Provides the earliest definitive genetic diagnosis, allowing more time for parental decision-making.
- Risks & Limitations:
- Slightly higher risk of fetal loss (~1%) vs. amniocentesis.
- ⚠️ Risk of transverse limb defects if performed before 10 weeks.
- Cannot detect Neural Tube Defects (NTDs).
- Potential for confined placental mosaicism, which may require follow-up amniocentesis.
⭐ High-Yield: Unlike amniocentesis, CVS does not analyze amniotic fluid. Therefore, it cannot detect open neural tube defects, which require measuring alpha-fetoprotein (AFP).
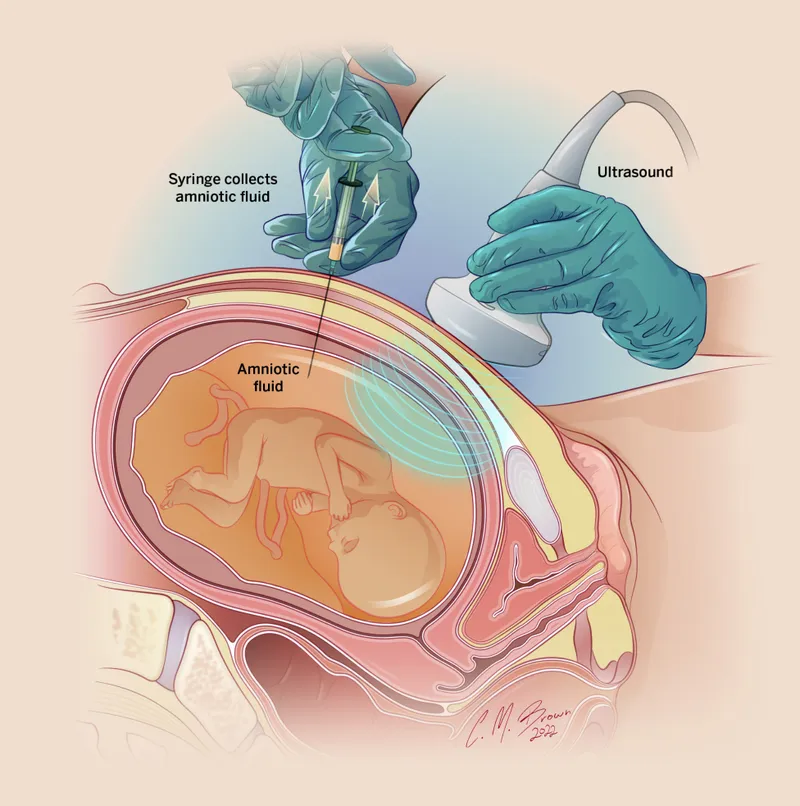
Amniocentesis - The Fluid Truth
-
Procedure: Transabdominal needle aspiration of amniotic fluid using ultrasound guidance, collecting fetal cells (amniocytes) for analysis.
-
Timing: Performed between 15-20 weeks gestation. Can be done later for fetal lung maturity assessment.
-
Indications:
- Advanced maternal age (≥35).
- Abnormal aneuploidy screening results (e.g., first-trimester screen, quad screen).
- Parental carrier of a chromosomal translocation.
- History of a child with a structural birth defect or chromosomal abnormality.
-
Risks:
- Procedure-related fetal loss rate is low, approx. 1/900.
- ⚠️ Potential for membrane rupture, infection, or needle injury to the fetus.
⭐ Amniotic fluid acetylcholinesterase (AChE) is a highly specific marker for open neural tube defects.
Procedural Face-Off - CVS vs. Amnio
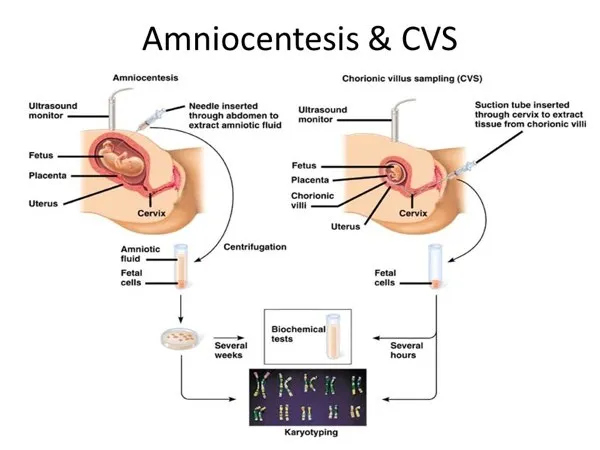
| Feature | Chorionic Villus Sampling (CVS) | Amniocentesis |
|---|---|---|
| Timing | 10-13 weeks | 15-20 weeks |
| Sample | Placental villi (chorion frondosum) | Amniotic fluid with fetal cells |
| Route | Transcervical or transabdominal | Transabdominal |
| Risk | Fetal loss (~1%); ↑ risk of limb reduction defects if <10 wks | Fetal loss (~0.1-0.3%) |
| Pros | Earlier diagnosis | Safer; can measure AFP |
| Cons | Cannot detect NTDs; risk of maternal cell contamination | Later diagnosis |
High‑Yield Points - ⚡ Biggest Takeaways
- Amniocentesis is performed at 15-20 weeks; Chorionic Villus Sampling (CVS) is done earlier, at 10-13 weeks.
- Only amniocentesis can detect neural tube defects by measuring alpha-fetoprotein (AFP).
- CVS has a slightly higher risk of fetal loss and can cause limb reduction defects if performed before 10 weeks.
- Amniocentesis samples fetal cells from amniotic fluid; CVS samples placental tissue.
- Confined placental mosaicism is a potential diagnostic pitfall specific to CVS.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

