Viral Blueprint - The Cancer Playbook
- Direct-acting: Viruses either carry a viral oncogene (v-onc) or integrate their genome near a cellular proto-oncogene (c-onc), causing overexpression (insertional mutagenesis).
- Indirect-acting: Viral proteins can bind to and inactivate cell cycle regulators and tumor suppressors.
- Chronic Inflammation: Persistent viral infection (e.g., HCV, HBV) causes chronic inflammation, leading to ↑ cell turnover and accumulation of mutations.
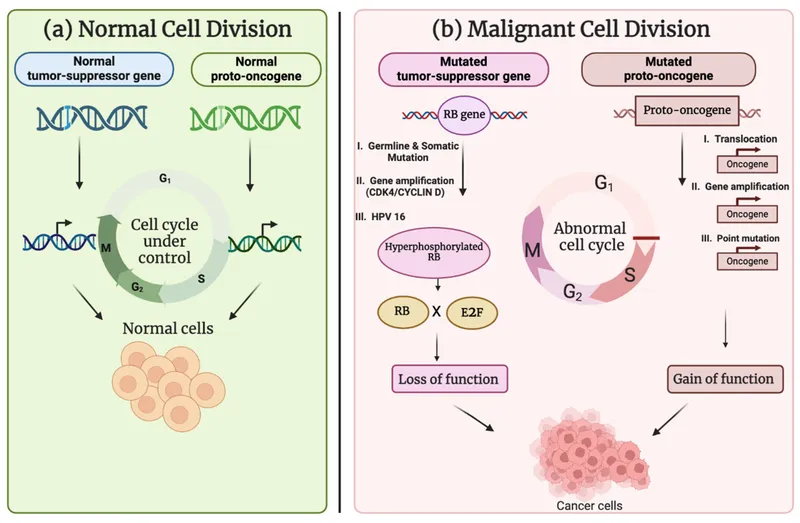
⭐ High-yield example: Human Papillomavirus (HPV) produces the E6 protein, which leads to the degradation of p53, and the E7 protein, which inhibits the Retinoblastoma (Rb) protein.
DNA Viruses - Cellular Hijackers
DNA viruses typically integrate their genome into host DNA. Their oncoproteins target and neutralize the cell's primary "brakes" on proliferation: the p53 and Rb tumor suppressor proteins. This forces the cell into a state of continuous replication, increasing the risk of malignant transformation.
-
Human Papillomavirus (HPV)
- High-risk serotypes: 16, 18, 31, 33.
- Viral Oncoproteins:
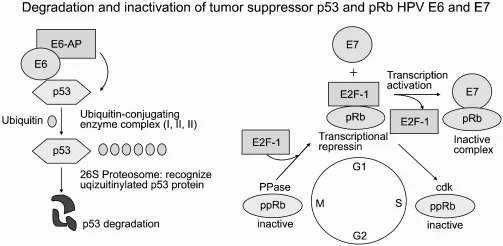
- $E6$: Promotes ubiquitin-mediated degradation of the p53 tumor suppressor.
- $E7$: Binds and inactivates the retinoblastoma (Rb) protein.
- Mechanism: Inactivating Rb releases E2F transcription factor, pushing the cell past the G1/S checkpoint. Loss of p53 prevents apoptosis in response to this abnormal growth signal.
-
Epstein-Barr Virus (EBV)
- Proteins: Latent Membrane Protein 1 (LMP-1) acts as a constitutively active CD40 receptor, promoting B-cell proliferation. EBNA-2 transactivates host proto-oncogenes like c-myc.
- Cancers: Burkitt Lymphoma, nasopharyngeal carcinoma.
-
Other Key DNA Viruses
- Hepatitis B (HBV): Primarily drives cancer via chronic inflammation and hepatocyte regeneration. The HBx protein can also transactivate growth-promoting genes.
- HHV-8 (KSHV): Encodes viral homologs of key cellular proteins (v-cyclin, v-BCL-2) that directly promote cell cycling and inhibit apoptosis.
⭐ High-risk HPV strains (16, 18) cause >70% of cervical cancers. The combined inactivation of both p53 (by $E6$) and Rb (by $E7$) is the critical driver of malignant transformation.
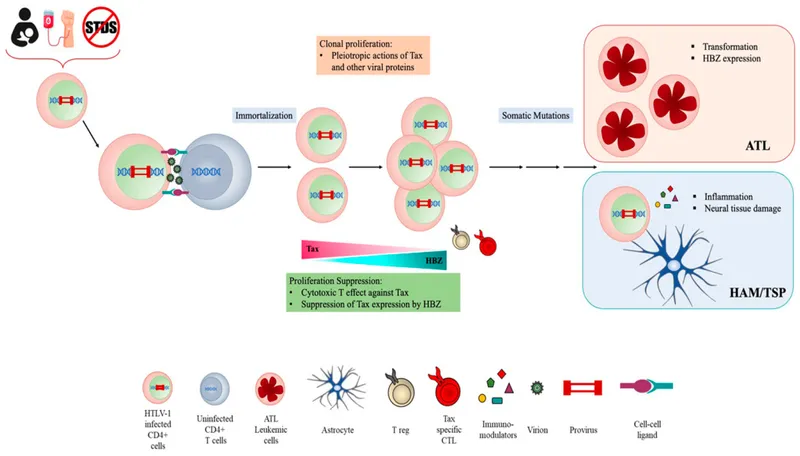
RNA Retroviruses - The Tax Man Cometh
- Human T-lymphotropic Virus (HTLV-1): A deltaretrovirus.
- Encodes the oncoprotein Tax, a key transcriptional trans-activator.
- Tax stimulates host cell proliferation by activating genes like $IL-2$ and its receptor, $IL-2R$.
- This drives polyclonal T-cell expansion, increasing the risk of secondary mutations.
- Mechanism: Indirect action; does not rely on insertional mutagenesis near a proto-oncogene.
- Associated Malignancy: Adult T-cell Leukemia/Lymphoma (ATLL).
⭐ HTLV-1 exhibits a long latency period (decades), with ATLL developing in only <5% of those chronically infected.
High‑Yield Points - ⚡ Biggest Takeaways
- Viral oncogenesis hinges on inactivating tumor suppressors (e.g., p53, Rb) and activating proto-oncogenes.
- HPV uses E6 to degrade p53 and E7 to inhibit Rb, forcing cell cycle progression.
- HBV and HCV induce hepatocellular carcinoma primarily via chronic inflammation and hepatocyte regeneration.
- EBV (Burkitt lymphoma) and HHV-8 (Kaposi's sarcoma) express oncoproteins that mimic host growth factors.
- HTLV-1's Tax protein transactivates host cell genes, driving proliferation in adult T-cell leukemia/lymphoma.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

