Initial Colonization - The First Roommates
- In Utero: Traditionally viewed as sterile, but low-level microbial DNA suggests some prenatal exposure.
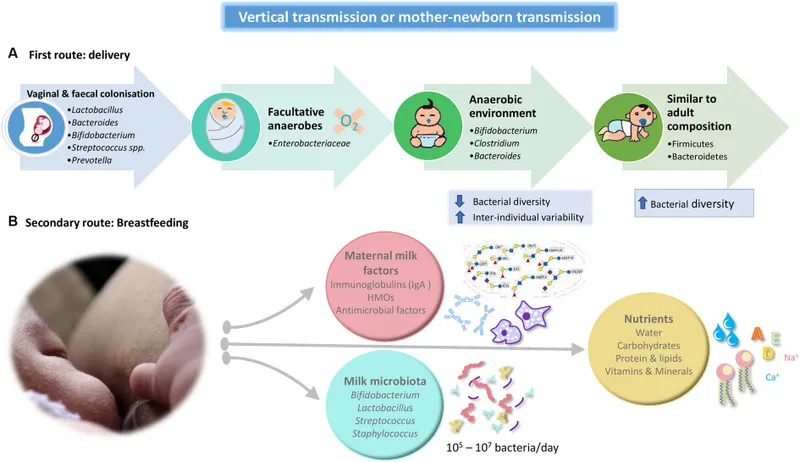
- At Birth (Primary Inoculation): Mode of delivery is the single most important factor.
- Vaginal Delivery: Infant gut colonized by maternal vaginal & fecal flora. Predominantly Lactobacillus, Prevotella, and Bacteroides. This initial profile is linked to a lower risk of atopic diseases.
- Cesarean Section: Colonized by maternal skin flora and the hospital environment. Primarily Staphylococcus, Corynebacterium, and Propionibacterium.
⭐ High-Yield: Infants born via C-section have delayed colonization with key gut commensals like Bacteroides and Bifidobacterium, which are critical for educating the neonatal immune system.
Postnatal Influences - Shaping the Community
-
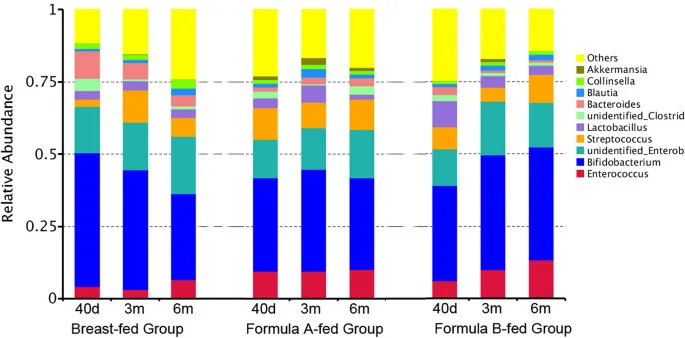
Diet: Breast vs. Formula
- Breast milk: Rich in human milk oligosaccharides (HMOs), which act as prebiotics.
- Promotes protective bacteria like Bifidobacterium and Lactobacillus.
- Associated with ↓ risk of infections and atopic disease.
- Formula milk: Lacks HMOs. Fosters a more diverse, adult-like microbiome earlier, with more Bacteroides and Clostridium.
- Breast milk: Rich in human milk oligosaccharides (HMOs), which act as prebiotics.
-
Environmental Factors
- Exposure: Contact with siblings, pets, and rural environments increases microbial diversity.
- Antibiotics: Broad-spectrum antibiotics disrupt the microbiome (dysbiosis), causing ↓ diversity and increasing long-term risk for obesity, IBD, and allergies.
⭐ Hygiene Hypothesis: Proposes that reduced microbial exposure in early life impairs immune system development, thereby increasing susceptibility to allergic and autoimmune diseases.
Microbiome Maturation - Growing Up Inside
- Initial Colonization: Fetus is largely sterile. Microbiome acquisition begins at birth, heavily influenced by delivery mode.
- Vaginal Birth: Colonized by maternal vaginal flora (e.g., Lactobacillus, Prevotella).
- C-section: Colonized by maternal skin & environmental microbes (e.g., Staphylococcus).
- Postnatal Factors:
- Diet: Human Milk Oligosaccharides (HMOs) in breast milk selectively promote Bifidobacterium growth.
- Environment & Antibiotics: Environmental exposures increase diversity, while antibiotics can disrupt maturation.
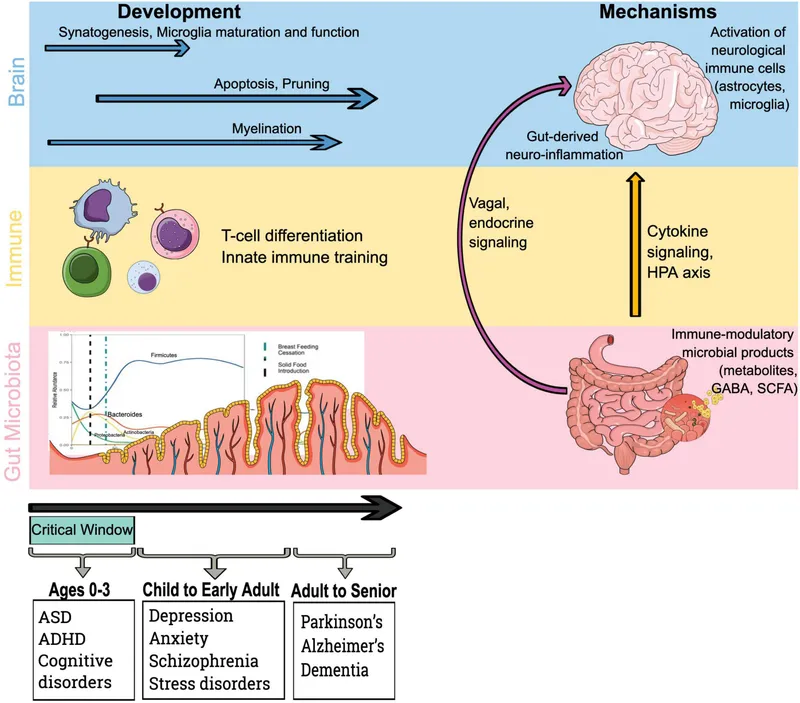
⭐ Altered infant microbiomes, such as from C-sections or antibiotic use, are linked to a higher risk of developing allergies, asthma, and obesity.
Clinical Correlations - When Flora Fails
- Dysbiosis: Disruption of normal flora, often by antibiotics (e.g., Clindamycin, Fluoroquinolones), creating an opportunity for pathogens.
- Key Opportunistic Pathogens:
- Clostridioides difficile: Spore-forming anaerobe; overgrowth leads to pseudomembranous colitis.
- Candida albicans: Fungal overgrowth causing oral thrush or vulvovaginitis.
- Bacteroides fragilis: Gut commensal; can cause intra-abdominal abscesses if displaced by trauma/surgery.
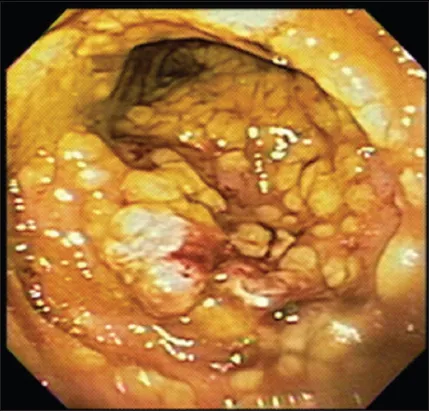
⭐ Exam Favorite: C. difficile Toxin A is an enterotoxin targeting brush border enzymes, causing watery diarrhea. Toxin B is a cytotoxin that disrupts the cytoskeleton, leading to necrosis and pseudomembrane formation.
High‑Yield Points - ⚡ Biggest Takeaways
- The uterus is sterile; initial microbial colonization occurs during birth.
- Vaginal delivery seeds the infant with maternal vaginal/fecal flora (Lactobacillus), while C-section seeds skin flora (Staphylococcus).
- Breastfeeding is crucial, promoting Bifidobacterium growth via human milk oligosaccharides (HMOs).
- The infant microbiome has low diversity, stabilizing to an adult-like state by age 2-3.
- Early-life dysbiosis is linked to future allergies, asthma, and autoimmune disorders.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

