Biofilm Basics - Microbial Slime Cities
- What: Structured communities of microbes encased in a self-produced, slimy Extracellular Polymeric Substance (EPS) matrix.
- Why: Adhere to surfaces, especially medical devices (catheters, prosthetic joints, heart valves), protecting bacteria from host defenses and antibiotics.
- Key Features:
- EPS Matrix: Polysaccharide shield blocks antibiotics & immune cells.
- Quorum Sensing: Bacteria communicate to coordinate defense, growth, and virulence.
- Slow Growth: Cells in deeper layers are metabolically inactive, resisting drugs that target growth.
⭐ Biofilms can be up to 1000x more resistant to antibiotics than their free-floating (planktonic) counterparts.
Infected Hardware - Rogues' Gallery
Biofilms colonize medical devices, forming a protective slime matrix that shields them from antibiotics and immune cells. This makes device removal a common necessity for cure.
- IV Catheters & Prosthetic Joints/Valves:
- Staphylococcus epidermidis (coagulase-negative)
- Staphylococcus aureus (coagulase-positive)
- Ventilator-Associated Pneumonia (VAP):
- Pseudomonas aeruginosa (produces green pigment)
- Acinetobacter baumannii
- Catheter-Associated UTI (CAUTI):
- E. coli
- Proteus mirabilis (urease-positive)
- Klebsiella pneumoniae
- Contact Lenses:
- Pseudomonas aeruginosa (can cause keratitis)
- CNS Shunts:
- S. epidermidis
⭐ The extracellular polymeric substance (EPS) matrix of biofilms-composed of polysaccharides, proteins, and eDNA-is the primary reason for the ↑1000x resistance to antibiotics compared to their planktonic counterparts.

Clinical Clues - The Silent Siege
- Insidious Onset: Infections are often low-grade, indolent, and lack classic systemic signs of sepsis. Think subacute presentation, not acute crisis.
- Device-Centered: Symptoms localize to the implanted device (e.g., joint pain, catheter site inflammation, new murmur with a prosthetic valve).
- Recalcitrant to Antibiotics: Standard antibiotic courses often fail or lead to transient improvement, with relapse after therapy stops. The biofilm acts as a shield.
- Culture Negativity: Planktonic (free-floating) bacteria may be cleared, leading to negative blood cultures despite an active, localized infection on the device surface.
⭐ Staphylococcus epidermidis, a normal skin commensal, is the most common cause of biofilm-related infections on prosthetic devices and IV catheters.
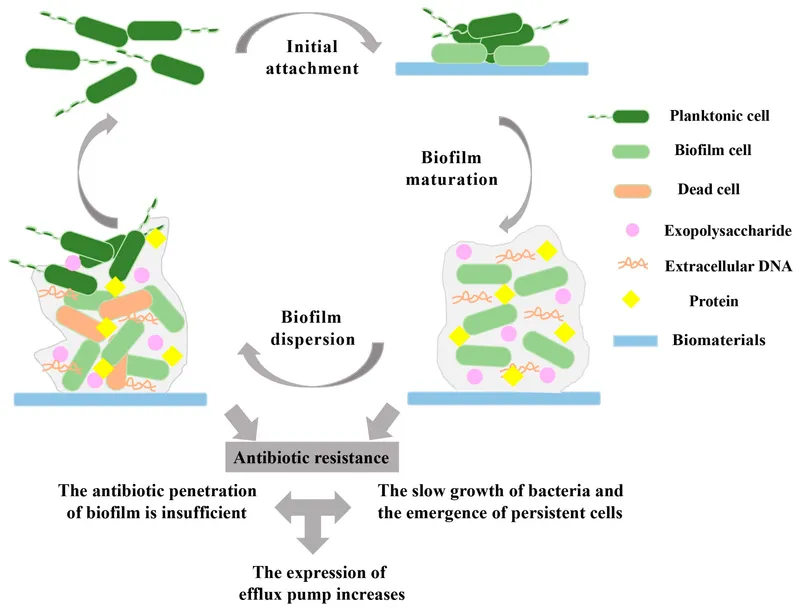
The Resistance - Breaking the Fortress
-
Resistance Mechanisms:
- Physical Barrier: The EPS matrix physically blocks or slows antibiotic penetration.
- Altered Microenvironment: ↓O₂ and nutrient gradients create dormant persister cells, which are metabolically inactive and thus tolerant to many antibiotics.
- Gene Regulation: Upregulation of efflux pumps and stress-response genes.
- Horizontal Gene Transfer: Close cell proximity facilitates the exchange of resistance plasmids.
-
Clinical Approaches:
- Source Control: Device removal is often the most critical step for cure.
- Pharmacotherapy: Use of high-dose, prolonged combination antibiotic regimens.
⭐ Persister cells within a biofilm are a major cause of treatment failure and infection recurrence. They are phenotypically tolerant to antibiotics due to metabolic dormancy, not genetic resistance.
High‑Yield Points - ⚡ Biggest Takeaways
- Biofilms are microbial communities encased in a self-produced extracellular polysaccharide (EPS) matrix.
- They are a major cause of device-related infections, colonizing catheters, prosthetic joints, and heart valves.
- Key pathogens include S. epidermidis, S. aureus, Pseudomonas aeruginosa, and Candida albicans.
- The matrix provides profound resistance to antibiotics and host immune defenses, leading to persistent infections.
- Bacteria within biofilms communicate via quorum sensing to regulate their collective behavior.
- Eradication is challenging; device removal is often the only definitive treatment.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

