Pathophysiology - Gland on the Fritz
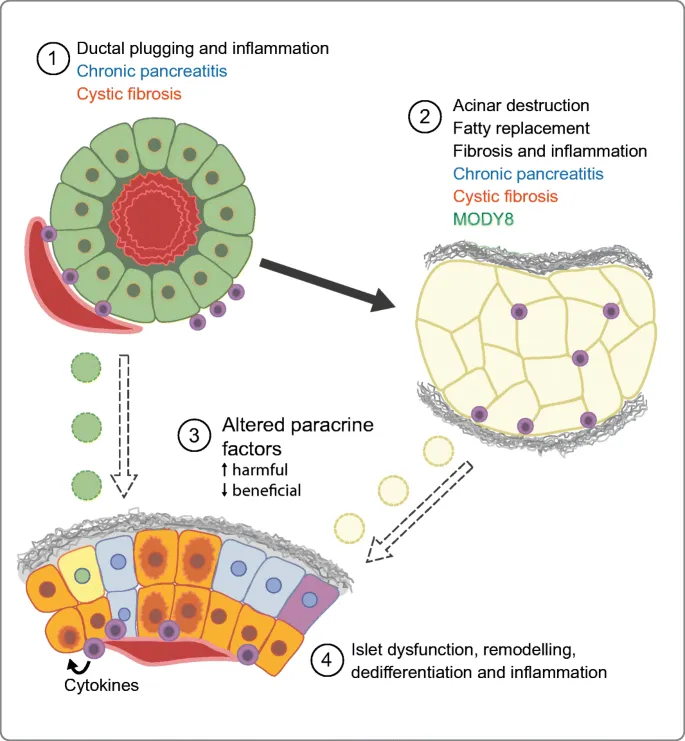
- Acute or chronic inflammation leads to enzymatic autodigestion and fibrotic replacement of pancreatic parenchyma, directly destroying the Islets of Langerhans.
- This process damages both β-cells (producing insulin) and α-cells (producing glucagon).
- The result is pancreatogenic or Type 3c diabetes mellitus (T3cDM).
- Unlike Type 1 or 2 DM, T3cDM involves a dual hormone deficit, creating a volatile metabolic state often called “brittle diabetes.”
⭐ Exam Favorite: The concurrent loss of glucagon (the primary counter-regulatory hormone to insulin) makes patients exceptionally sensitive to exogenous insulin, leading to a high risk of severe, unpredictable hypoglycemia.

Diabetes Mellitus (Type 3c) - The Brittle Aftermath
-
Pathophysiology: Arises from irreversible damage to the endocrine pancreas (islets of Langerhans) due to inflammation, fibrosis, or surgical resection in chronic or severe acute pancreatitis. It's a pancreatogenic or secondary diabetes.
-
Key Defect: Involves concurrent loss of both insulin-producing β-cells and glucagon-producing α-cells. This dual hormone deficiency leads to:
- Hyperglycemia: Due to insulin lack.
- Brittle Glycemic Control: Marked by a high risk of severe, unpredictable hypoglycemia because of the impaired glucagon counter-regulatory response.
-
Clinical & Diagnostic Features:
- Often coexists with exocrine insufficiency (maldigestion, steatorrhea).
- Patients typically have lower insulin requirements than in Type 1 DM.
- Absence of typical autoimmune markers seen in Type 1 DM (e.g., anti-GAD antibodies).
-
Management:
- Requires cautious insulin therapy due to hypoglycemia risk.
- Pancreatic Enzyme Replacement Therapy (PERT) is crucial for malabsorption and can help stabilize blood glucose.
⭐ In Type 3c diabetes, iatrogenic hypoglycemia is a major clinical threat because the body's primary defense, glucagon release from α-cells, is compromised along with insulin production.
Diagnosis & Management - Walking the Tightrope
- Diagnosis: Monitor blood glucose (BG) frequently. Differentiate stress hyperglycemia from new-onset post-pancreatitis diabetes mellitus (T3cDM).
- Management: A careful balance to avoid hypoglycemia.
- Target BG: 140-180 mg/dL for most hospitalized patients.
- Insulin Therapy: IV insulin infusion in ICU; subcutaneous basal-bolus regimen preferred over sliding scale for others. Avoid oral agents in acute illness.
⭐ Pancreatic diabetes (T3cDM) is often "brittle," with unpredictable glycemic swings and high hypoglycemia risk due to concomitant glucagon deficiency from α-cell destruction.
High‑Yield Points - ⚡ Biggest Takeaways
- Acute pancreatitis can cause transient hyperglycemia due to stress and cytokine release.
- Chronic pancreatitis leads to pancreatogenic diabetes (Type 3c) from progressive, irreversible islet cell destruction.
- The key pathology is the loss of both insulin (beta cells) and glucagon (alpha cells).
- This results in "brittle diabetes," characterized by extreme glycemic lability.
- A hallmark is the high risk of severe hypoglycemia due to a deficient glucagon response.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

