RCT Fundamentals - The Gold Standard
- Definition: The gold standard experimental design to establish causality between an intervention and an outcome.
- Core Principle: Compares outcomes in a group receiving an intervention against a control group receiving a placebo or standard of care.
- Key Features:
- Randomization: Allocates subjects to groups by chance.
- Blinding: Prevents bias; can be single, double, or triple-blind.
⭐ RCTs are the most effective study design for minimizing confounding by randomly distributing potential confounders between groups.
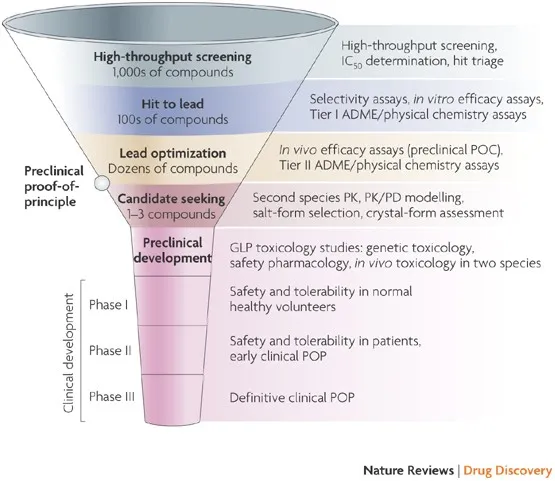
Phases of Clinical Trials - The Drug Gauntlet
📌 Mnemonic: SWIM
- Safe? (Phase I)
- Works? (Phase II)
- Improvement? (Phase III)
- Market? (Phase IV)
| Phase | Purpose | Participants |
|---|---|---|
| I | Safety & Toxicity ("Is it safe?") | ~20-80 healthy volunteers |
| II | Efficacy & Dosing ("Does it work?") | ~100s with disease |
| III | Compare to Standard of Care ("Is it better?") | ~1000s, multiple centers (RCT) |
| IV | Post-marketing surveillance | General population |
Bias & Confounding - Taming the Gremlins
-
Bias: Systematic error skewing results from the true value.
- Selection Bias: Non-random assignment. Prevented by randomization.
- Performance Bias: Differences in care between groups. Prevented by blinding of participants/personnel.
- Detection Bias: Outcome assessment differs between groups. Prevented by blinding of outcome assessors.
- Attrition Bias: Unequal loss to follow-up. Mitigated by intention-to-treat (ITT) analysis.
-
Confounding: A third variable associated with both exposure and outcome distorts the apparent relationship.
- Controlled during design by randomization, restriction, and matching.
⭐ Confounding vs. Effect Modification: Confounding is a bias to be removed. Effect modification is a real finding to be described and reported.
Analysis Principles - The Final Verdict
- Intention-to-Treat (ITT): "Once randomized, always analyzed."
- Includes all subjects in their originally assigned group, regardless of adherence or withdrawal.
- Preserves randomization, avoids bias, and reflects real-world effectiveness.
- Per-Protocol (PP):
- Includes only subjects who completed the trial according to the protocol.
- Estimates treatment efficacy in a perfect scenario but is susceptible to selection bias.
⭐ ITT is the primary analysis for most RCTs because it provides an unbiased estimate of the treatment effect in a practical setting.
High‑Yield Points - ⚡ Biggest Takeaways
- Randomization is the most critical element, minimizing selection bias and confounding.
- Blinding (masking) prevents performance bias (participants/staff) and detection bias (observers).
- Intention-to-treat analysis includes all randomized patients, preserving randomization and reflecting real-world effectiveness.
- Per-protocol analysis evaluates only compliant patients, showing efficacy under ideal conditions but risking bias.
- A control group (placebo or standard of care) is essential for comparison.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

