Adaptive Designs - Plan B for Trials
- Allows for pre-planned modifications to a trial based on interim data analysis, offering flexibility over fixed designs.
- Primary Goals: Increase trial efficiency, reduce costs, and improve ethical outcomes by stopping futile trials early.
- Common Adaptations:
- Sample size re-estimation (SSR).
- Stopping early for futility or overwhelming efficacy.
- Dropping or adding treatment arms.
- ⚠️ Requires complex statistical adjustments (e.g., alpha-spending functions) to prevent inflating Type I error.
⭐ The most frequent adaptation is Sample Size Re-estimation (SSR). It uses interim data to recalculate the required sample size, ensuring the study remains adequately powered.
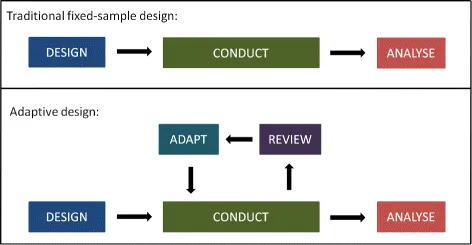
Sample Size Re-estimation - Sizing It Up
- An adaptive trial design method allowing for sample size adjustment during the study.
- Performed at a pre-planned interim analysis based on accumulating data.
- Primary Goal: To correct initial, potentially inaccurate, assumptions about effect size or data variability, ensuring adequate statistical power.
- Blinded Re-estimation: Adjusts sample size based on variance without unblinding treatment assignments. Lower risk of bias.
- Unblinded Re-estimation: Uses interim treatment effects to adjust size. More powerful but carries a higher risk of operational bias.
⭐ Unblinded sample size re-estimation can inflate the Type I error rate (false positives) if not controlled by specific statistical methods (e.g., promising zone design).
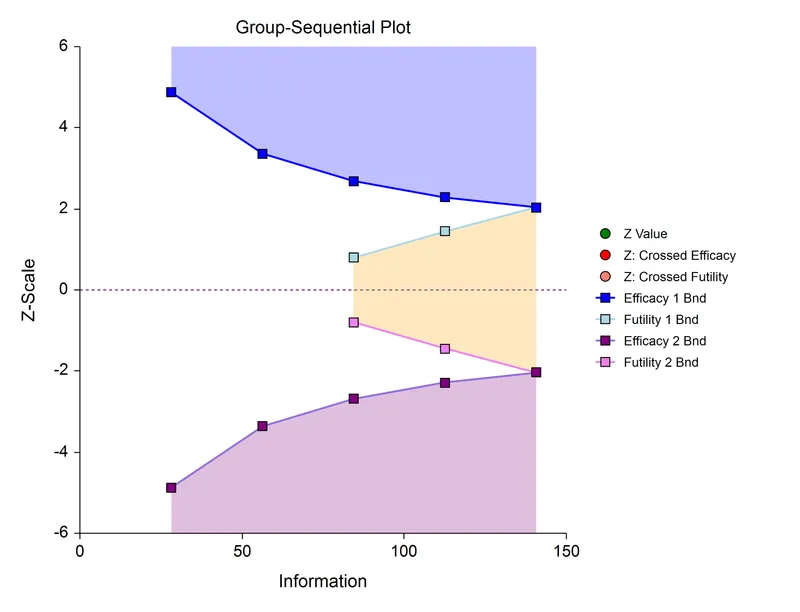
Group Sequential Designs - Peek-a-Boo Trials
- Concept: Allows for planned interim analyses of accumulating data in a clinical trial. The trial can be stopped early if results are compelling for efficacy, futility, or safety.
- Mechanism:
- Data is analyzed at pre-specified time points or after a certain number of events.
- Formal stopping rules (boundaries) are used to determine if the trial should continue.
- Challenge & Solution:
- ⚠️ Repeatedly "peeking" at the data inflates the overall Type I error rate (family-wise error rate).
- To maintain the nominal overall $\alpha$ (e.g., 0.05), the significance level for each interim look must be adjusted downwards.
- Common Boundary Methods:
- Pocock: Uses the same significance boundary for each interim analysis.
- O'Brien-Fleming: Uses conservative boundaries early on, making it hard to stop, and less conservative ones later.
⭐ High-Yield: The O'Brien-Fleming method is often preferred as it maintains statistical power better than the Pocock method by being very conservative at the initial interim looks.
- Adaptive designs allow for pre-planned sample size recalculation mid-trial based on interim data.
- This ensures the study remains adequately powered if initial effect size or variability estimates were inaccurate.
- The primary goal is to improve trial flexibility and resource efficiency.
- All adaptation rules must be prospectively defined in the study protocol to prevent bias.
- Helps avoid underpowered studies (↑ Type II error) or unnecessarily exposing patients in overpowered studies.
- Maintains statistical validity and blinding when executed properly.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

