Protein Folding - The Shape of Life
- Primary (1°): Linear amino acid sequence, determined by mRNA code.
- Secondary (2°): Localized structures like α-helices and β-pleated sheets, stabilized by hydrogen bonds.
- Tertiary (3°): Overall 3D shape of one polypeptide chain, driven by hydrophobic interactions, ionic bonds, and disulfide bridges.
- Quaternary (4°): Combination of two or more polypeptide subunits (e.g., hemoglobin).

- Chaperones (Heat Shock Proteins, e.g., Hsp60/Hsp70): Cellular proteins that assist in the proper folding of other proteins and prevent aggregation.
⭐ Protein misfolding and aggregation is a key pathological mechanism in neurodegenerative diseases, such as amyloid-β plaques in Alzheimer's disease and α-synuclein in Parkinson's disease.
Chaperones - Folding Supervisors
- Function: Proteins that assist in the proper folding of nascent or denatured polypeptides, preventing their aggregation. They don't determine the final structure.
- Mechanism: Bind to exposed hydrophobic regions on unfolded proteins in an ATP-dependent process.
- Heat Shock Proteins (HSPs): A key family of chaperones whose expression is upregulated during cellular stress (e.g., heat, toxins).
- HSP70: Binds to polypeptide chains as they emerge from the ribosome.
- HSP60 (Chaperonins): Form a barrel-shaped complex that encloses an unfolded protein, providing an isolated environment to fold.

⭐ High-Yield Fact: Proteins that cannot be refolded by chaperones are tagged by ubiquitin for degradation via the proteasome. Defects in this system are implicated in diseases like Alzheimer's and Parkinson's disease.
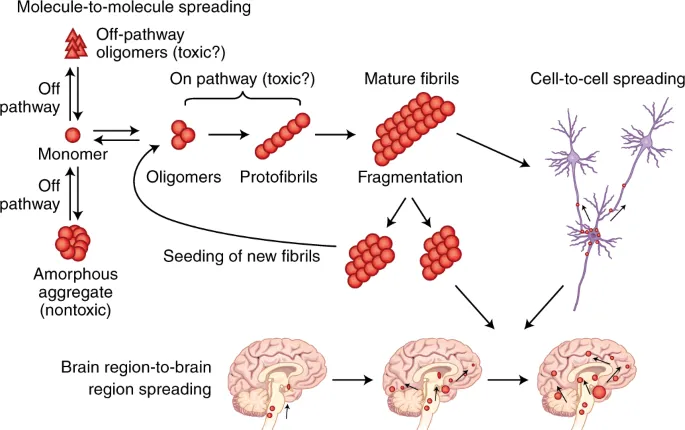
Misfolding Pathologies - When Good Proteins Go Bad
- Protein misfolding can lead to aggregation, forming insoluble amyloid fibrils or plaques that are resistant to degradation.
- These aggregates are often rich in β-pleated sheet structures.
- Pathology arises from either loss-of-function of the correctly folded protein or a toxic gain-of-function from the aggregate.
Key Amyloid Diseases:
- Alzheimer's Disease: Extracellular plaques of Amyloid-β (Aβ) and intracellular neurofibrillary tangles of hyperphosphorylated Tau protein.
- Parkinson's Disease: Aggregates of α-synuclein form intracellular Lewy bodies.
- Huntington's Disease: Trinucleotide (CAG) repeat expansion results in a polyglutamine tract in the huntingtin protein, causing aggregation.
- Prion Disease (e.g., CJD): Misfolded prion protein (PrPsc) catalyzes the conformational conversion of normal PrPc.
⭐ Prion diseases are uniquely transmissible; the misfolded PrPsc protein acts as an infectious agent, inducing misfolding in native PrPc proteins.
Ubiquitin-Proteasome System - Cellular Cleanup Crew
- Function: Degrades damaged or unnecessary proteins, preventing their toxic accumulation.
- Process (ATP-Dependent):
- Tagging: Proteins are marked for destruction by a polyubiquitin chain.
- Requires a cascade of three enzymes: E1 (activating), E2 (conjugating), and E3 (ligase).
- E3 ligases provide specificity by recognizing the target protein.
- Degradation: The 26S proteasome complex recognizes the ubiquitin tag, unfolds the protein, and chops it into small peptides.
- Tagging: Proteins are marked for destruction by a polyubiquitin chain.
⭐ Proteasome inhibitors (e.g., Bortezomib) are used to treat multiple myeloma by causing toxic protein buildup in cancer cells, leading to apoptosis.

High‑Yield Points - ⚡ Biggest Takeaways
- Protein folding dictates function; the primary amino acid sequence determines the final 3D conformation.
- Chaperone proteins (e.g., Hsp60, Hsp70) use ATP to facilitate correct folding and prevent aggregation.
- Misfolded proteins can accumulate, leading to diseases like Alzheimer (β-amyloid), Parkinson (α-synuclein), and prion diseases (PrPsc).
- The ubiquitin-proteasome system tags and degrades misfolded proteins in an ATP-dependent manner.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

