Core Principles - The Belmont Trio
Established by the Belmont Report, this framework guides US human research ethics.
- Respect for Persons: Treat individuals as autonomous agents.
- Requires robust informed consent: full disclosure, comprehension, and voluntary participation.
- Mandates special protections for those with diminished autonomy (e.g., children, prisoners, cognitively impaired).
- Beneficence: An obligation to "do no harm" (non-maleficence) and to maximize benefits while minimizing risks.
- Justice: Fair distribution of the burdens and benefits of research.
- Demands equitable selection of subjects, preventing exploitation of vulnerable groups.
⭐ Vulnerable populations (e.g., children, prisoners, pregnant women) lack full autonomy and require additional protections to prevent coercion or undue influence.
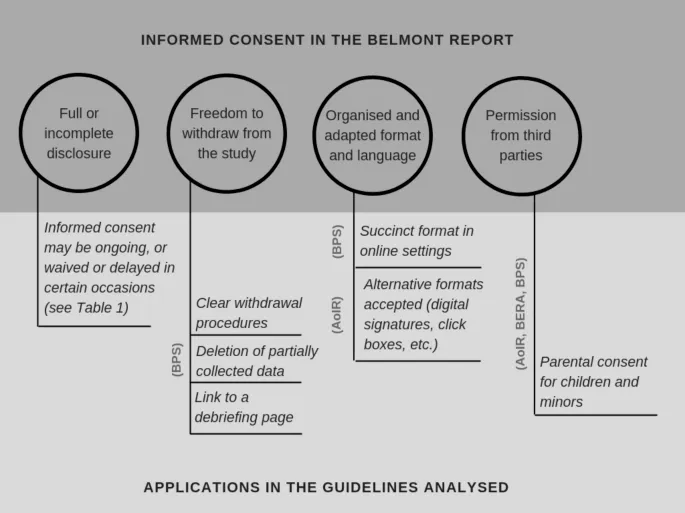
Informed Consent - Getting the Nod
-
Core Principle: An ongoing, voluntary dialogue ensuring participants understand a study's purpose, procedures, potential risks, benefits, and alternatives before agreeing. It is a process, not a single event.
-
Pillars of Valid Consent:
- Disclosure: Imparting all relevant information. 📌 Mnemonic: BRAIN (Benefits, Risks, Alternatives, Indications, Nature of procedure).
- Understanding: Ensuring the participant comprehends the information; requires clear, jargon-free language.
- Capacity: The individual's ability to make an autonomous, rational decision.
- Voluntariness: Freedom from coercion or undue influence.
-
Exceptions in Research:
- Waiver of consent is possible for minimal-risk research via IRB approval.
- Emergency research has strict, narrow exceptions.
⭐ For minors, obtain informed consent from parents/guardians and assent (affirmative agreement) from the child, typically if aged >7 years.
Institutional Review Board - The Ethics Police
- Formal committee that approves, monitors, and reviews biomedical and behavioral research involving humans.
- Primary mandate: Protect the rights, safety, and welfare of human research subjects.
- Composition: Minimum 5 members with diverse backgrounds (scientific, non-scientific, and community members).
- Reviews protocols for ethical concerns, risk-benefit analysis, and informed consent procedures.
⭐ To ensure a balanced perspective, the IRB must include at least one non-scientist and one member unaffiliated with the institution.
Vulnerable Subjects - Handle With Care
- Require extra protections against coercion or undue influence; the IRB must implement additional safeguards.
- Key populations include:
- Children & minors
- Prisoners
- Pregnant women, fetuses, and neonates
- Cognitively impaired or educationally/economically disadvantaged individuals
- 📌 For children, obtain parental/guardian permission AND the child's own assent (agreement), if they are capable (e.g., age >7).
- For prisoners, incentives must not be coercive (e.g., suggesting parole).
⭐ For research involving children, both parental permission and child assent are ethically required. Assent is the child's affirmative agreement, not just a failure to object.
High‑Yield Points - ⚡ Biggest Takeaways
- Informed consent is mandatory; subjects must be competent and understand the study.
- Beneficence (do good) and non-maleficence (do no harm) are paramount.
- Justice requires fair selection of subjects, avoiding exploitation of vulnerable populations.
- Autonomy respects a patient's right to make their own decisions about participation.
- All research requires Institutional Review Board (IRB) approval to protect human subjects.
- Confidentiality of patient data must be maintained throughout the study.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

