Hemostasis - The First Plug
- Initiation: Vessel injury exposes subendothelial collagen and von Willebrand Factor (vWF).
- Step 1: Vasoconstriction
- A transient neurogenic reflex, reinforced by endothelin.
- Immediately reduces blood flow.
- Step 2: Primary Hemostasis (Platelet Plug)
- Adhesion: Platelet receptor GpIb binds to vWF on exposed collagen.
- Activation: Platelets change shape and degranulate, releasing ADP and Thromboxane A₂ ($TXA_2$).
- Aggregation: Fibrinogen cross-links platelets via the GpIIb/IIIa receptor, forming the initial plug.
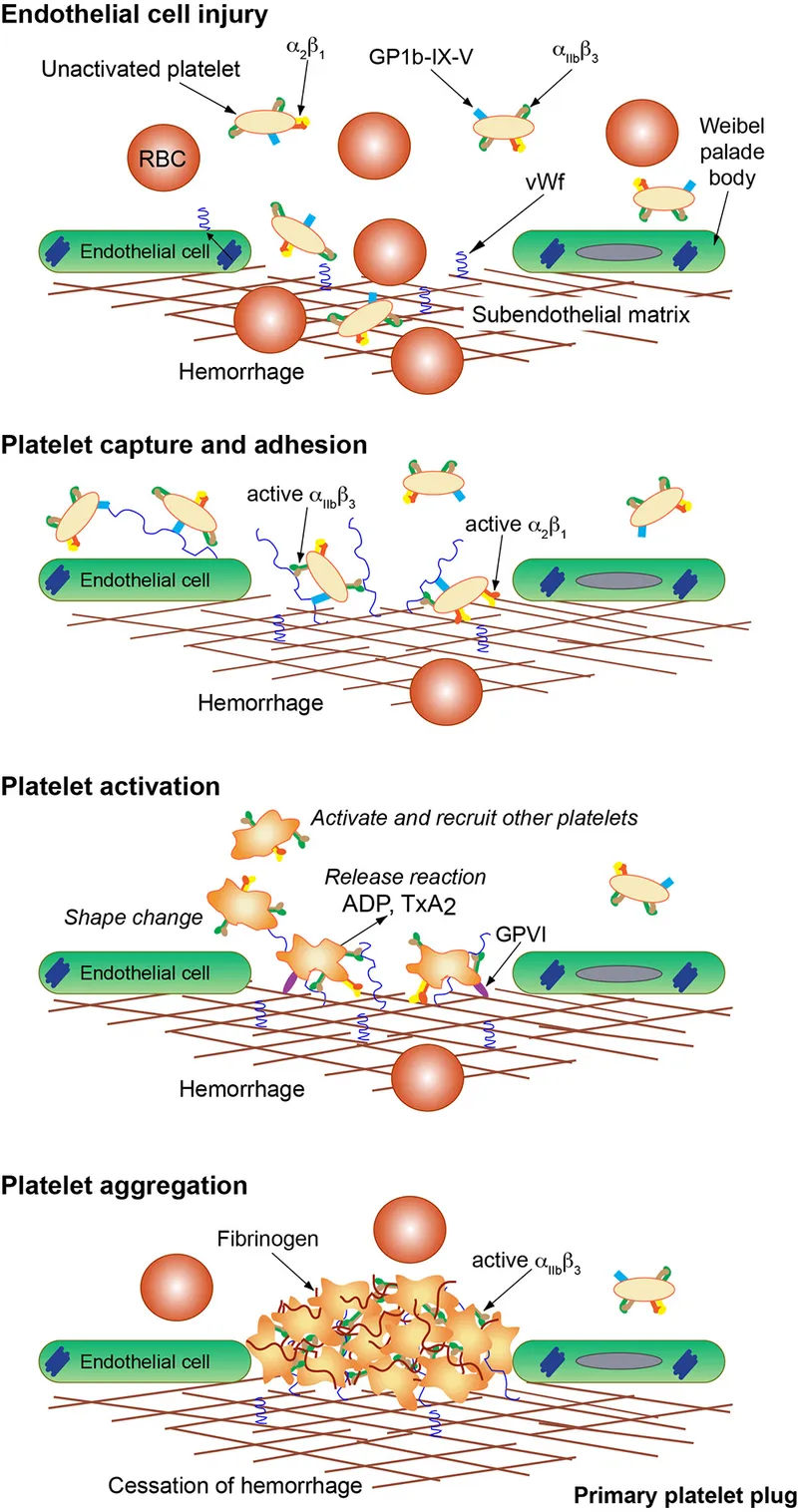
⭐ Glanzmann Thrombasthenia is an autosomal recessive disorder caused by a deficiency of GpIIb/IIIa, leading to impaired platelet aggregation and a severe bleeding diathesis.
Inflammation - The Clean‑Up Crew
- Timeline: Day 0-3, initiated by the coagulation cascade and complement activation.
- Vascular Response: Transient vasoconstriction (hemostasis) is followed by vasodilation and increased permeability.
- Cellular Influx:
- Neutrophils (PMNs): Peak at 24-48 hours. They are the primary defense against infection, performing phagocytosis of bacteria and debris.
- Macrophages (Monocytes): Become dominant by 48-72 hours. They are essential for debridement and orchestrate the transition to proliferation by releasing growth factors (PDGF, TGF-β, VEGF).
- 📌 Mnemonic: Neutrophils Navigate, Macrophages Manage.
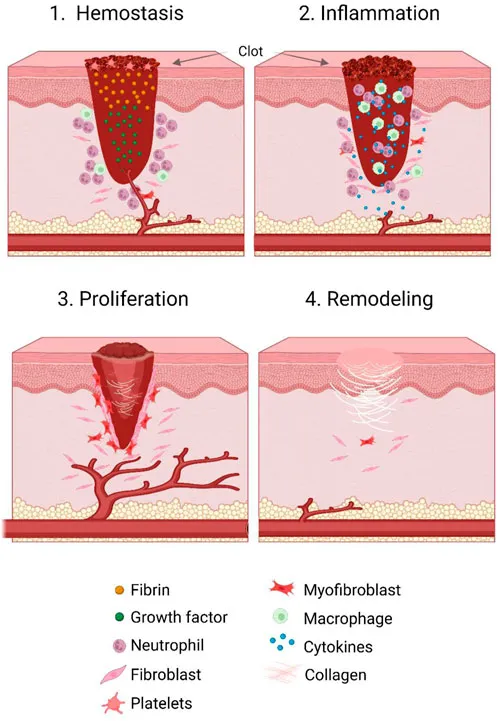
⭐ Macrophages are the critical directors of wound repair. Their depletion results in impaired debridement and a complete failure to initiate the proliferative phase, leading to a chronic wound state.
Proliferation - The Rebuilding
- Timeline: Day 3 to weeks.
- Hallmark: Formation of pink, soft granulation tissue.
- Fibroplasia: Fibroblasts (driven by PDGF, FGF, TGF-β) migrate and deposit ground substance and Type III collagen.
- Angiogenesis: New capillary formation (driven by VEGF, FGF) gives the tissue its granular look.
- Epithelialization: Keratinocytes migrate from wound edges to cover the surface.
- Wound Contraction:
- Myofibroblasts (specialized fibroblasts with actin filaments) contract, shrinking the wound.
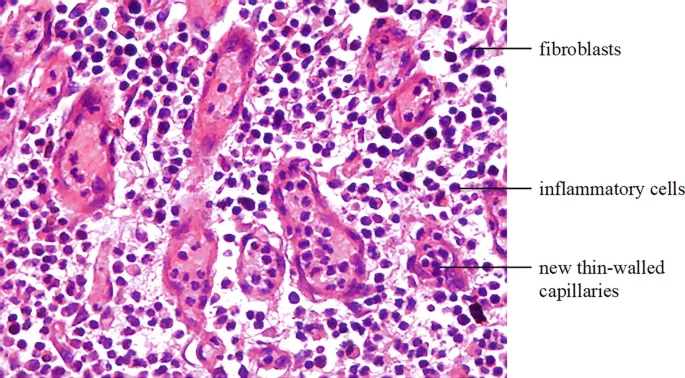
⭐ The initial scaffold is made of Type III collagen. This is later replaced by stronger Type I collagen in the remodeling phase, a critical concept for understanding scar strength.
Remodeling - The Final Polish
- Timeline: Begins week 3; can last for 1-2 years.
- Primary Goal: ↑ Tensile strength through collagen reorganization.
- Type III collagen is replaced by stronger Type I collagen.
- Key enzymes: Matrix Metalloproteinases (MMPs) degrade collagen; Lysyl oxidase cross-links it.
- Tensile Strength:
- Plateaus at ~80% of the original tissue's strength after several months.
- Clinical Appearance: The scar flattens, softens, and pales over time.
- Pathology: Imbalance in collagen synthesis/degradation leads to hypertrophic scars or keloids.
⭐ The ratio of Type I to Type III collagen is a key indicator of wound maturity. A mature scar approaches the normal skin ratio of approximately 4:1.
High‑Yield Points - ⚡ Biggest Takeaways
- Inflammatory phase: Neutrophils arrive first, but macrophages are critical for transitioning to the proliferative phase.
- Proliferative phase: Defined by granulation tissue formation, with fibroblasts depositing Type III collagen and myofibroblasts causing contraction.
- Remodeling phase: Type III collagen is replaced by stronger Type I collagen, a process that can last for more than a year.
- Macrophages are the most critical cell type, directing the entire repair process.
- Final wound strength is at most ~80% of the original tissue.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

