🧬 The Widening Wall
- Focal aortic dilation > 3.0 cm or >50% of normal diameter; typically infrarenal.
- Pathophysiology: Chronic inflammation → ↑Matrix Metalloproteinases (MMPs) → degradation of elastin & collagen → wall weakening.
- Key Risks: Smoking (strongest), male sex, age >65, family history, atherosclerosis.
⭐ Law of Laplace ($T \propto P \times r$): As AAA radius (r) increases, wall tension (T) increases, driving further expansion and ↑ rupture risk.
🛡️ Pathophysiology - Wall Under Siege
- Chronic Inflammation: Infiltration of macrophages & T-cells into the aortic wall.
- Proteolytic Degradation: ↑ Matrix Metalloproteinases (MMPs) & elastases degrade elastin and collagen.
- Structural Failure: Leads to smooth muscle cell apoptosis and loss of wall integrity.
- Law of Laplace: Wall tension $T \propto P \times r$. As radius (r) ↑, tension (T) ↑, promoting expansion.
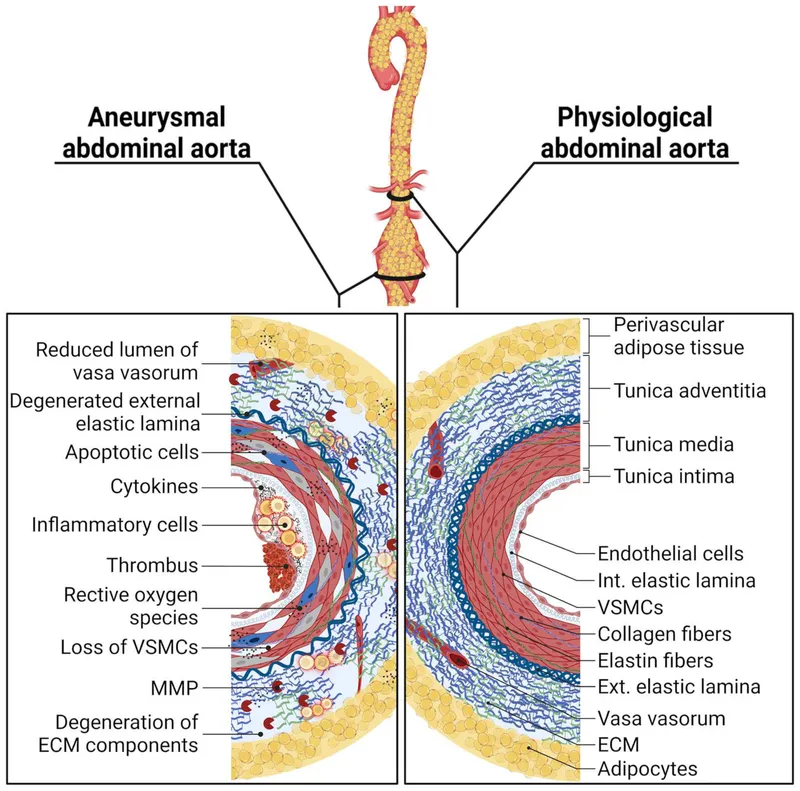
⭐ Location: Most AAAs are infrarenal, likely due to reduced vasa vasorum supply, leading to relative ischemia and wall weakness.
🤫 Clinical Manifestations - The Silent Killer
-
Asymptomatic (Most Common):
- Usually an incidental finding on imaging.
- May present as a palpable, pulsatile abdominal mass.
-
Symptomatic (Non-ruptured):
- Constant, gnawing abdominal, flank, or back pain.
- Suggests rapid expansion or impending rupture.
-
Ruptured AAA (Classic Triad):
- Sudden, severe pain.
- Hypotension/shock.
- Pulsatile mass.
⭐ The classic triad of rupture (pain, hypotension, pulsatile mass) is present in only ~50% of patients.
📏 Diagnosis - Sizing Up the Threat
- Initial Test & Screening: Abdominal Ultrasound (US).
- Pre-op Planning (Gold Standard): CT Angiography (CTA).
- Defines anatomy: diameter, length, neck morphology, renal/iliac artery involvement.
- 💡 Aortic diameter > 3.0 cm is defined as an aneurysm.
⭐ Repair is indicated for aneurysms > 5.5 cm in men, > 5.0 cm in women, or those with rapid growth (>0.5 cm in 6 months or >1 cm/year).
🔪 Management: Open Repair vs. EVAR
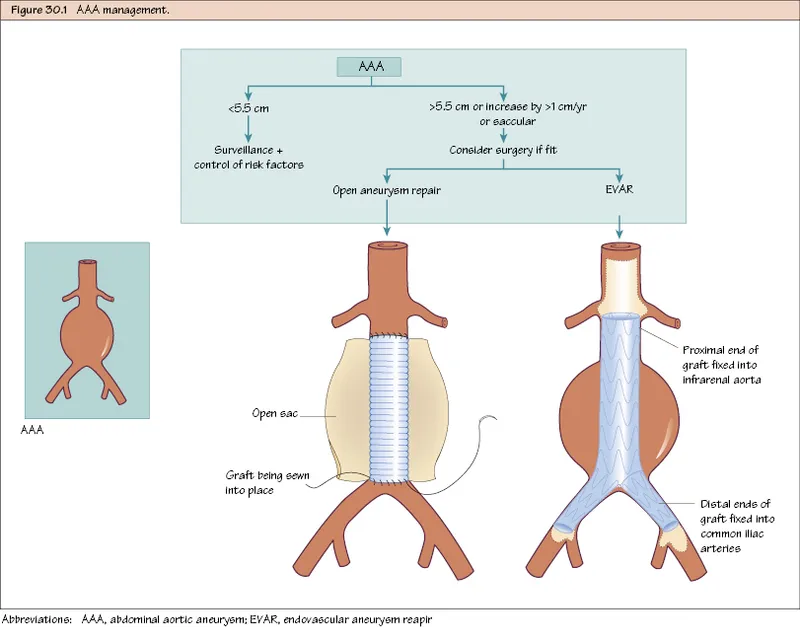
| Feature | Open Surgical Repair (OSR) | Endovascular Aneurysm Repair (EVAR) |
|---|---|---|
| Approach | Midline laparotomy, aortic cross-clamp, sutured graft | Percutaneous femoral access, deployed stent-graft |
| Mortality | Higher peri-op mortality (3-5%) | Lower peri-op mortality (<1.5%) |
| Recovery | Longer hospital stay, ↑ pain | Shorter hospital stay, ↓ pain |
| Late Complication | Aortoenteric fistula, graft infection | Endoleak, graft migration/fracture |
| Follow-up | Less intensive surveillance | Lifelong imaging surveillance required |
⭐ > EVAR's primary long-term complication is endoleak: persistent blood flow into the aneurysm sac outside the stent-graft. Type II is most common (from lumbar/IMA back-bleeding) and often observed. Type I (inadequate seal) requires urgent intervention.
💥 Complications - The Aftermath
- Early (<30d): MI (most common cause of death), AKI, colon ischemia (IMA ligation), graft thrombosis, limb ischemia.
- Late (>30d): Aortoenteric fistula (herald bleed → massive GI bleed), graft infection (S. aureus), anastomotic pseudoaneurysm.
- EVAR-Specific: Endoleak (persistent blood flow into aneurysm sac), graft migration.
⭐ Bowel ischemia classically presents with bloody diarrhea and abdominal pain post-op, often involving the sigmoid colon (watershed area of IMA).
⚡ Biggest Takeaways
- Screening is a one-time ultrasound for men 65-75 who have ever smoked.
- Repair is indicated for diameter >5.5 cm, rapid growth (>0.5 cm/6 mo), or symptoms.
- The classic triad of rupture is hypotension, a pulsatile abdominal mass, and severe back/flank pain.
- EVAR has lower perioperative mortality but requires surveillance for endoleaks.
- Open repair risks include ischemic colitis (early) and aortoenteric fistula (late).
- Myocardial infarction is the leading cause of death post-operatively.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

