Acid-Base Basics - The Body's pH Tango
- pH: Critical measure of acidity. Normal range: 7.35-7.45.
- < 7.35 = Acidosis
-
7.45 = Alkalosis
- Regulation: A delicate balance maintained by two main organs:
- Lungs (Respiratory): Rapidly adjust $PaCO_2$ (acid). Normal: 35-45 mmHg.
- Kidneys (Metabolic): Slowly regulate $HCO_3^-$ (base). Normal: 22-26 mEq/L.
- Governing Principle: The Henderson-Hasselbalch equation: $pH \propto \frac{[HCO_3^-]}{PaCO_2}$.
⭐ Exam Favourite: Acidosis causes extracellular potassium ($K^+$) excess (hyperkalemia) as $H^+$ ions move into cells, forcing $K^+$ out. Alkalosis does the opposite, causing hypokalemia.
Metabolic Acidosis - Sour Grapes
- Primary Disturbance: Low pH from a primary decrease in serum bicarbonate ($HCO_3^-$). Compensatory response is hyperventilation to decrease $pCO_2$.
- Key Diagnostic Step: Calculate the anion gap (AG) to narrow the differential diagnosis.
- Formula: $AG = [Na^+] - ([Cl^-] + [HCO_3^-])$
- Normal AG is < 12 mEq/L.
- High AG (HAGMA) Causes: 📌 MUDPILES: Methanol, Uremia, DKA, Propylene glycol, Isoniazid, Lactic acidosis, Ethylene glycol, Salicylates.
- Normal AG (NAGMA) Causes: 📌 HARDUPS: Hyperalimentation, Acetazolamide, RTA, Diarrhea, Uretero-enteric fistula, Pancreatic fistula, Saline.
⭐ In surgical patients, NAGMA is commonly caused by loss of bicarbonate-rich fluids. Think diarrhea, a high-output ileostomy, or a pancreatic/biliary fistula.
Metabolic Alkalosis - Basic Instinct
- Etiology: Most common acid-base disorder in surgical patients, from loss of H⁺ or gain of bicarbonate ($HCO_3^-$).
- Key Causes: Vomiting, nasogastric (NG) suction, diuretic therapy, massive blood transfusion (citrate metabolism).
- Diagnosis: ABG shows pH > 7.45, ↑ PaCO₂, ↑ $HCO_3^-$. Urine chloride (UCl) is key to management.
⭐ Paradoxical Aciduria: With severe hypokalemia, kidneys excrete H⁺ instead of K⁺ to conserve potassium, causing acidic urine despite systemic alkalosis.
Respiratory Rollercoaster - Huffing & Puffing
📌 ROME: Respiratory is Opposite (↑$pCO_2$ leads to ↓pH; ↓$pCO_2$ leads to ↑pH).
-
Respiratory Acidosis (Hypoventilation)
- Primary Problem: Inadequate alveolar ventilation causing $CO_2$ retention.
- Surgical Causes: Post-op atelectasis, CNS depression (opioids, anesthesia), airway obstruction, severe pneumonia.
- Labs: pH < 7.35, $pCO_2$ > 45 mmHg.
- Compensation: Slow renal $HCO_3^-$ retention.
-
Respiratory Alkalosis (Hyperventilation)
- Primary Problem: Excessive $CO_2$ elimination.
- Surgical Causes: Pain/anxiety (most common), sepsis, iatrogenic over-ventilation.
- Labs: pH > 7.45, $pCO_2$ < 35 mmHg.
- Compensation: Slow renal $HCO_3^-$ excretion.
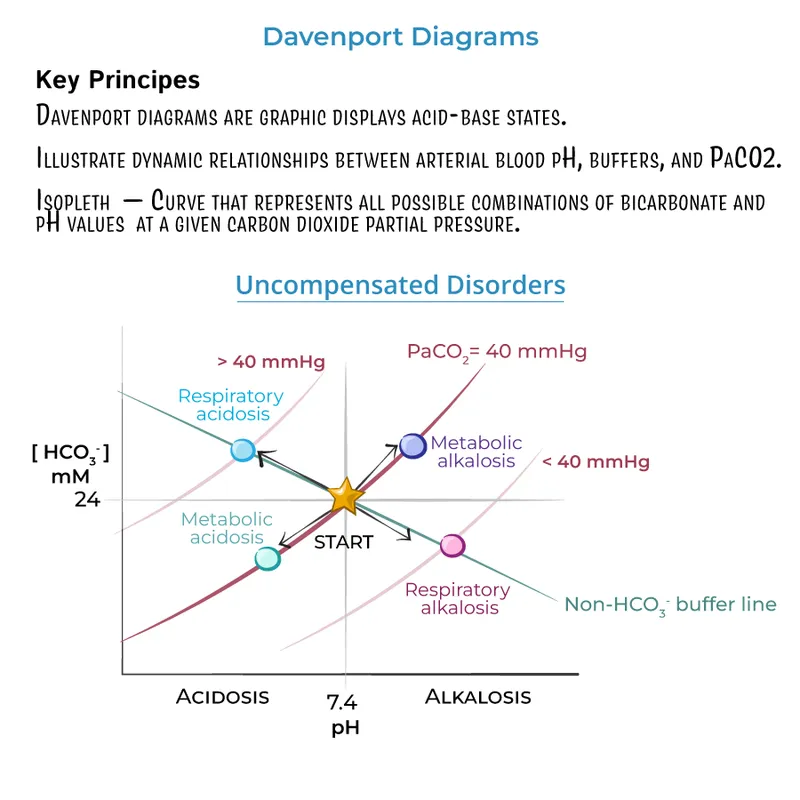
⭐ In acute respiratory acidosis, expect $HCO_3^-$ to increase by 1 mEq/L for every 10 mmHg rise in $pCO_2$. In chronic states (>48h), it compensates much more, rising by 3-4 mEq/L.
High-Yield Points - ⚡ Biggest Takeaways
- Metabolic acidosis with an elevated anion gap suggests lactic acidosis from shock or sepsis.
- Metabolic alkalosis, the most common disorder, is typically from NG tube suction or vomiting and is saline-responsive.
- Respiratory acidosis stems from hypoventilation (e.g., opioids, airway obstruction).
- Respiratory alkalosis is caused by hyperventilation (e.g., pain, anxiety, mechanical over-ventilation).
- Always treat the underlying cause revealed by the ABG, not just the numbers.
- Lungs compensate rapidly (minutes-hours); kidneys compensate slowly (days).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

