Gastric Phase - The Acid Test
- Stimulus: Vagal input & stomach distension trigger gastrin release.
- HCl Secretion: Parietal cells secrete HCl, lowering gastric pH to ~1.5-3.5.
- Pepsinogen Activation: Chief cells release inactive pepsinogen.
⭐ Pepsin is an endopeptidase that preferentially cleaves peptide bonds after aromatic amino acids (e.g., Phenylalanine, Tyrosine).
Intestinal Lumen - Pancreatic Power-up
Chyme entering the duodenum triggers the release of pancreatic zymogens (inactive proenzymes) to prevent pancreatic autodigestion. Their activation is a critical, cascaded event.
- Primary Activation: Brush border enzyme enteropeptidase (enterokinase) converts trypsinogen to its active form, trypsin.
- Cascade: Trypsin then activates other pancreatic zymogens.
- Endopeptidases: Trypsin, chymotrypsin, elastase cleave internal peptide bonds.
- Exopeptidases: Carboxypeptidases A & B cleave amino acids from the C-terminus.
- Result: Large proteins are broken down into smaller, absorbable oligopeptides, dipeptides, and tripeptides.
⭐ High-Yield Fact: Enteropeptidase, a brush border enzyme, is the master switch for activating protein digestion in the intestine. Its deficiency leads to severe protein malabsorption and failure to thrive.
Brush Border & Absorption - The Final Gateway
-
Final Digestion: Occurs at the apical membrane of enterocytes. Brush border enzymes hydrolyze small peptides into absorbable units.
- Enzymes: Aminopeptidases and dipeptidases cleave oligopeptides into amino acids, dipeptides, and tripeptides.
-
Absorption into Enterocyte:
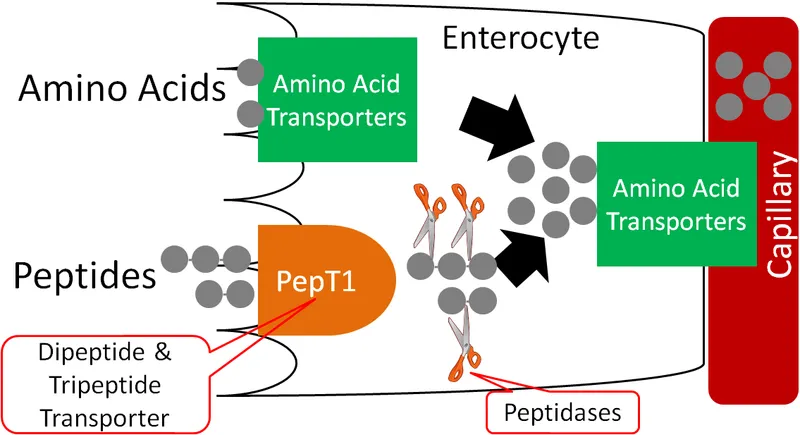
- Amino Acids: Absorbed via sodium-dependent co-transporters (e.g., B⁰AT1 for neutral AAs).
- Di- & Tripeptides: Absorbed faster via a proton-dependent co-transporter, PepT1.
- Inside the cell, most di/tripeptides are hydrolyzed to amino acids by cytoplasmic peptidases.
-
Exit to Blood: Amino acids exit the enterocyte via facilitated diffusion across the basolateral membrane into the portal circulation.
⭐ Hartnup Disease: An autosomal recessive defect in the B⁰AT1 transporter for neutral amino acids (e.g., Tryptophan). Leads to pellagra-like symptoms (dermatitis, dementia, diarrhea) due to deficient niacin synthesis.
Clinical Correlates - Pathway Pathologies
- Pancreatic Insufficiency
- Causes: Chronic pancreatitis, cystic fibrosis, pancreatic cancer.
- Mechanism: ↓ synthesis & secretion of pancreatic proteases (trypsin, chymotrypsin).
- Result: Protein malabsorption, azotorrhea (excess fecal nitrogen), and edema.
- Congenital Enteropeptidase Deficiency
- Rare autosomal recessive disorder preventing trypsinogen activation.
- Leads to severe, generalized protein maldigestion.
- Presents in infancy with failure to thrive, diarrhea, and hypoalbuminemia-induced edema.
- Amino Acid Transport Defects
- Hartnup Disease: Defective neutral amino acid (tryptophan) transporter in intestinal/renal cells.
⭐ Causes pellagra-like symptoms (dermatitis, dementia, diarrhea) due to impaired niacin (B3) synthesis from tryptophan.
- Cystinuria: Defective dibasic amino acid transporter (Cysteine, Ornithine, Lysine, Arginine 📌 COLA).
- Intestinal defect is clinically silent; main issue is recurrent cystine kidney stones from renal transport failure.
- Hartnup Disease: Defective neutral amino acid (tryptophan) transporter in intestinal/renal cells.
High‑Yield Points - ⚡ Biggest Takeaways
- Protein digestion begins in the stomach with pepsin; most occurs in the small intestine.
- Pancreatic proteases (trypsin, chymotrypsin) are secreted as inactive zymogens.
- Brush border enterokinase activates trypsinogen to trypsin, initiating an activation cascade.
- Final products are amino acids, dipeptides, and tripeptides, absorbed by specific transporters.
- Na+-dependent cotransport is the primary mechanism for amino acid absorption.
- Hartnup disease is a key pathology involving defective neutral amino acid transport.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

