O₂-Hgb Curve - The S-Shaped Star
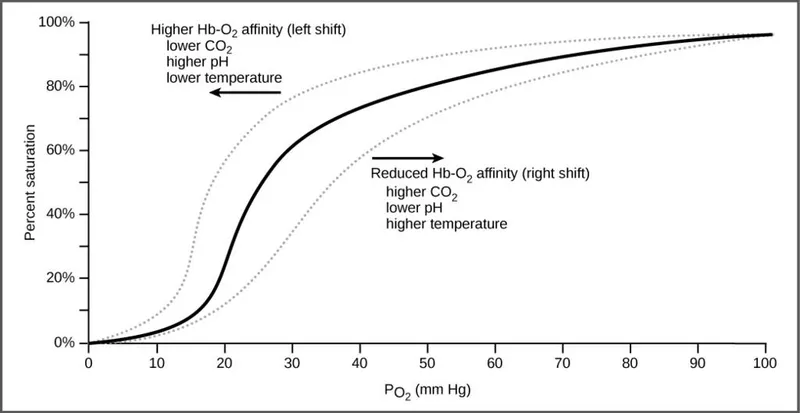
- Sigmoid (S-shape) curve due to cooperative binding: O₂ binding to one heme group ↑ affinity of others.
- P₅₀: O₂ partial pressure where hemoglobin is 50% saturated; normally ~26 mmHg.
| Right Shift (↓ Affinity) | Left Shift (↑ Affinity) |
|---|---|
| ↑ CO₂, ↑ Acid (↓ pH) | ↓ CO₂, ↓ Acid (↑ pH) |
| ↑ 2,3-DPG, ↑ Temp | ↓ 2,3-DPG, ↓ Temp |
| ↑ Exercise | Fetal Hb (HbF), CO, MetHb |
⭐ Bohr Effect: In peripheral tissues, ↑ H⁺ and ↑ CO₂ promote O₂ release from hemoglobin, causing a rightward shift of the curve.
Curve Shifts - Righty Tighty, Lefty Loosey
The curve's position reflects hemoglobin's O₂ affinity. A right shift promotes O₂ unloading in tissues (lower affinity), while a left shift enhances O₂ binding in the lungs (higher affinity).
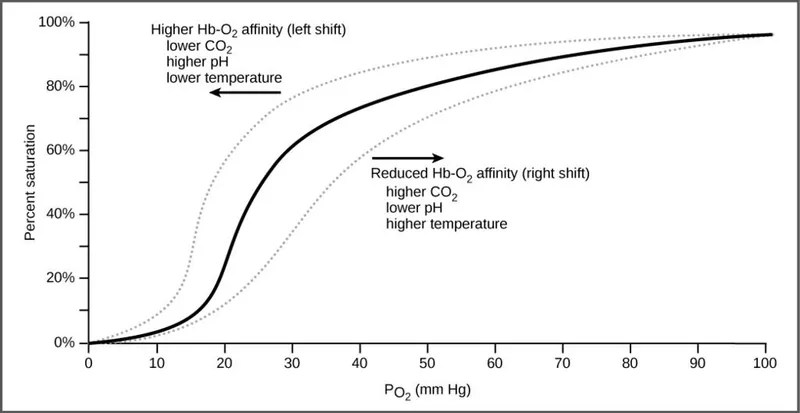
| Feature | Right Shift (↓ Affinity) | Left Shift (↑ Affinity) |
|---|---|---|
| P₅₀ | ↑ P₅₀ | ↓ P₅₀ |
| Causes | ↑ CO₂, ↑ Acid/H⁺ (↓pH), ↑ 2,3-DPG, ↑ Temperature, Exercise | ↓ CO₂, ↓ Acid/H⁺ (↑pH), ↓ 2,3-DPG, ↓ Temperature, Fetal Hemoglobin (HbF), CO, Methemoglobinemia |
| Mnemonic | 📌 CADET, face Right! (CO₂, Acid, 2,3-DPG, Exercise, Temperature) |
Clinical Curves - When Hemoglobin Misbehaves
- Curve Shifts: Changes in hemoglobin's affinity for oxygen ($O_2$).
- Left Shift: ↑ Affinity (Loves $O_2$). Less $O_2$ delivered to tissues.
- Right Shift: ↓ Affinity (Releases $O_2$). More $O_2$ delivered to tissues.
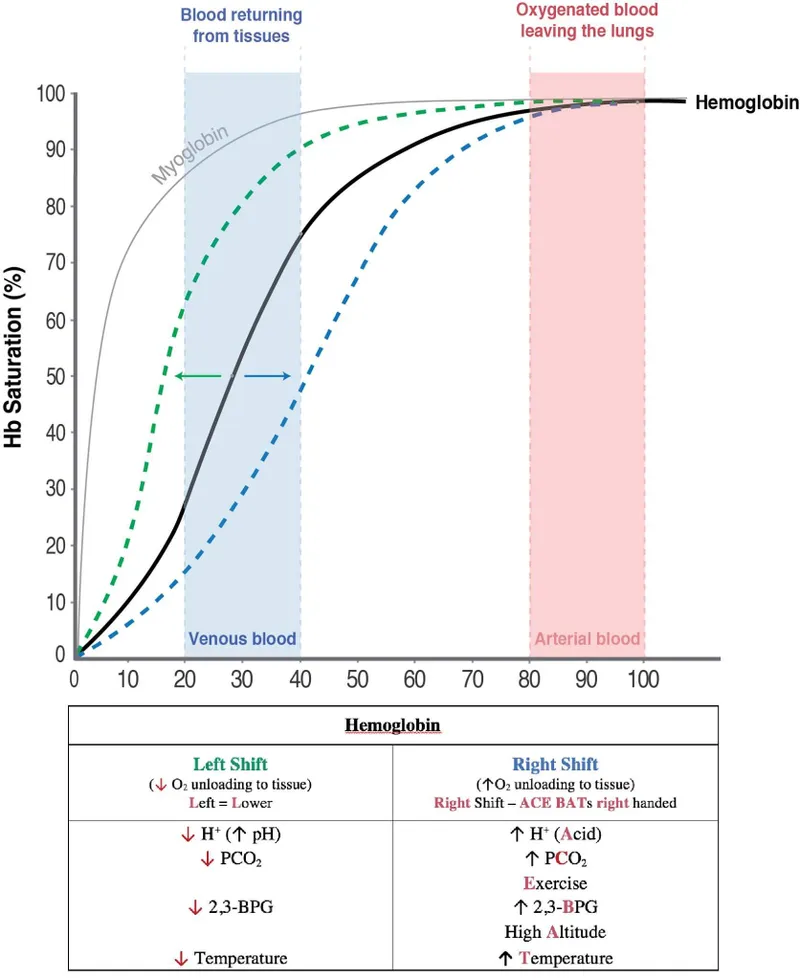
| Left Shift (↑ Affinity) | Factor | Right Shift (↓ Affinity) |
|---|---|---|
| ↓ $H^+$ (↑ pH) | Acidity | ↑ $H^+$ (↓ pH) |
| ↓ Temp | Temp. | ↑ Temp |
| ↓ 2,3-BPG | 2,3-BPG | ↑ 2,3-BPG |
| ↑ CO | CO | |
| ↑ Met-Hb | Methemoglobin | |
| ↑ HbF | Fetal Hb |
⭐ In CO poisoning, $PaO_2$ is normal, but $SaO_2$ is low. CO causes a left shift, trapping $O_2$ on hemoglobin.
- The sigmoidal shape of the curve is due to cooperative binding.
- A right shift indicates decreased O₂ affinity, facilitating O₂ unloading to tissues.
- Factors causing a right shift (CADET): ↑ CO₂, Acid (↓pH), 2,3-DPG, Exercise, Temperature.
- A left shift signifies increased O₂ affinity, promoting O₂ loading in the lungs.
- Key causes of a left shift include fetal hemoglobin (HbF) and CO poisoning.
- P50 is the PO₂ where hemoglobin is 50% saturated; an ↑ P50 reflects a right shift.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

