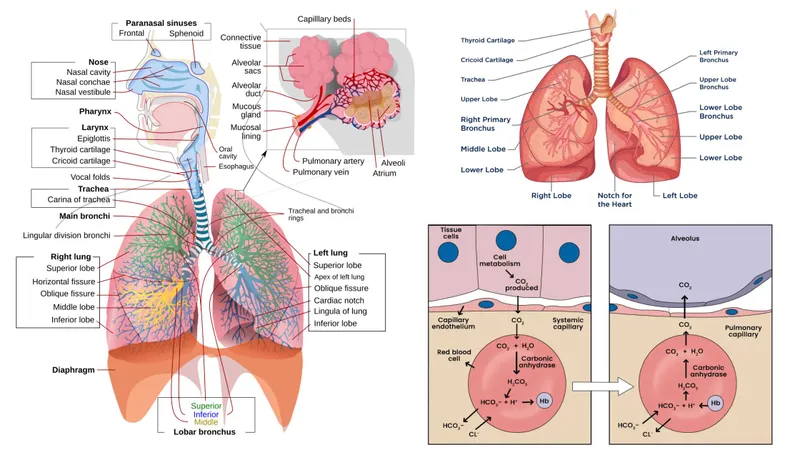
Chloride Shift - The Great CO₂ Exchange
- Mechanism: In tissues, CO₂ enters red blood cells (RBCs) and is converted to bicarbonate ($HCO_3^−$) and $H^+$.
- The Shift: To maintain electrical neutrality, $HCO_3^−$ is transported out of the RBC into the plasma in exchange for a chloride ion ($Cl^−$) moving into the RBC. This is facilitated by the Band 3 protein (anion exchanger 1).
- Haldane Effect: Deoxygenated hemoglobin has a higher affinity for $CO_2$ and $H^+$, facilitating uptake in tissues.
📌 Mnemonic: "Chloride Shifts for Carbonate" - $Cl^−$ shifts in to allow $HCO_3^−$ (carbonate) to shift out.
⭐ Exam Favorite: In the lungs, the process reverses. As $O_2$ binds to hemoglobin, $H^+$ is released, driving the conversion of bicarbonate back into $CO_2$, which is then exhaled. This is known as the reverse chloride shift.
Peripheral Tissues - The 'Hamburger' Effect
- Metabolically active tissues produce $CO_2$, which diffuses into Red Blood Cells (RBCs).
- Inside the RBC, the enzyme carbonic anhydrase rapidly converts $CO_2$ and $H_2O$ into carbonic acid ($H_2CO_3$).
- $CO_2 + H_2O \rightleftharpoons H_2CO_3 \rightleftharpoons H^+ + HCO_3^-$
- $H_2CO_3$ dissociates into a proton ($H^+$) and a bicarbonate ion ($HCO_3^-$).
- $H^+$ binds to deoxyhemoglobin, promoting $O_2$ release (Bohr effect).
- Bicarbonate ($HCO_3^-$) is transported out of the RBC into the plasma.
- To maintain electrical neutrality, a chloride ion ($Cl^-$) is transported into the RBC for every bicarbonate ion that exits. This is the chloride shift.
⭐ The chloride-bicarbonate exchange is facilitated by the Band 3 protein, also known as Anion Exchanger 1 (AE1). Defects can lead to hereditary spherocytosis.
Pulmonary Capillaries - The Reverse Shift
- In the lungs, high $O_2$ tension promotes oxygen binding to hemoglobin, leading to the release of $H^+$ ions (Haldane Effect).
- This reverses the chloride shift process seen in peripheral tissues.
- Released $H^+$ combines with bicarbonate ($HCO_3^-$) that moves into the RBC.
- Carbonic Anhydrase rapidly converts the resulting $H_2CO_3$ into $H_2O$ and $CO_2$.
- ↑ Intracellular $CO_2$ concentration drives its diffusion from the RBC into the alveoli for exhalation.
- To facilitate this, the Cl⁻/HCO₃⁻ exchanger (Band 3 protein) pumps $HCO_3^-$ into the RBC while pumping $Cl^-$ out.
⭐ Haldane Effect: Oxygenation of blood in the lungs displaces $CO_2$ from hemoglobin by decreasing its affinity for $CO_2$. This offloading is crucial for efficient $CO_2$ removal.
Clinical Correlations - Shift Storm
-
Systemic acid-base disorders directly impact the chloride shift by altering plasma pH and ion gradients, influencing gas transport efficiency.
-
Carbonic Anhydrase Inhibitors (e.g., Acetazolamide):
- Block the formation of carbonic acid ($H_2CO_3$) from $CO_2$ and $H_2O$ inside the RBC.
- This ↓ production of $H^+$ and $HCO_3^-$, impairing the chloride shift and $CO_2$ transport from tissues.
- Leads to a non-anion gap metabolic acidosis.
-
Diuretics (Loop & Thiazide):
- Can induce hypochloremic metabolic alkalosis.
- Renal loss of $Cl^-$ leads to compensatory ↑ in plasma $HCO_3^-$, altering the trans-membrane gradient.
⭐ In Salicylate (Aspirin) toxicity, a classic mixed acid-base disorder occurs: an initial respiratory alkalosis (from medullary stimulation) is followed by a high anion gap metabolic acidosis, disrupting the normal gas exchange equilibrium.
High‑Yield Points - ⚡ Biggest Takeaways
- The chloride shift (Hamburger phenomenon) is crucial for CO₂ transport in the blood.
- It exchanges intracellular bicarbonate (HCO₃⁻) for plasma chloride (Cl⁻) across the RBC membrane.
- This is facilitated by the Band 3 anion exchanger protein.
- In peripheral tissues, this allows the majority of CO₂ to be transported as bicarbonate in the plasma.
- The process reverses in the lungs, allowing CO₂ to be exhaled.
- This maintains the electrical neutrality of the RBC.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

