2,3-DPG Basics - The Oxygen Liberator
- Source: An organic phosphate created in red blood cells during anaerobic glycolysis via the Rapoport-Luebering shunt.
- Action: Binds to the beta chains of deoxygenated hemoglobin, stabilizing it in the low-affinity taut (T) state.
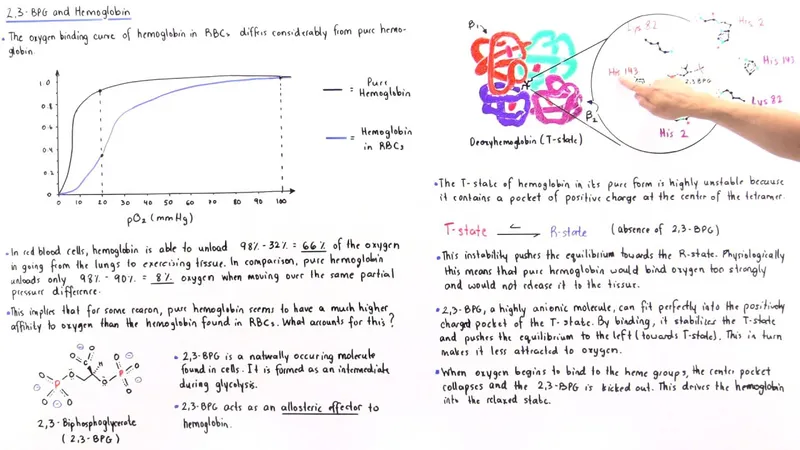
- Effect: Reduces hemoglobin's affinity for oxygen, promoting $O_2$ release to the tissues (a rightward shift of the dissociation curve).
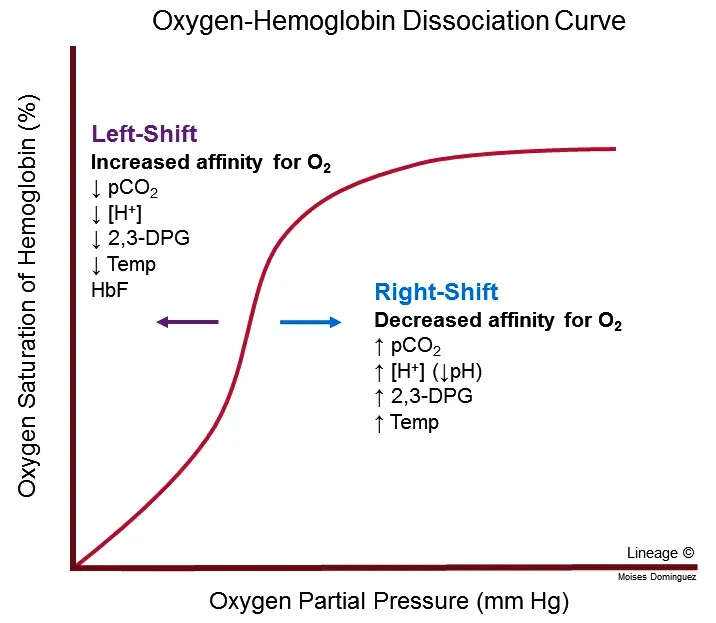
⭐ Fetal hemoglobin (HbF) has a low affinity for 2,3-DPG, granting it a higher oxygen affinity than adult hemoglobin (HbA) to facilitate oxygen uptake from the placenta.
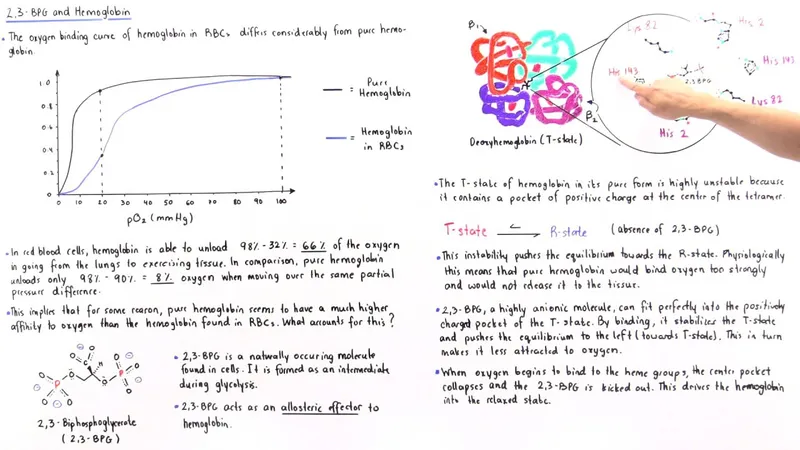
Mechanism of Action - The Great Let-Go
- Binding: 2,3-DPG fits into a pocket in the center of deoxygenated hemoglobin, binding to the two beta chains.
- Stabilization: This ionic binding stabilizes the low-affinity T (taut) state of hemoglobin.
- Effect: By locking hemoglobin in the T state, it reduces its affinity for oxygen.
- Outcome: Promotes the release of oxygen into the tissues, resulting in a rightward shift of the oxygen-hemoglobin dissociation curve.
⭐ In chronic hypoxia (e.g., high altitude, COPD) and anemia, RBCs ↑ 2,3-DPG production. This is a key compensatory mechanism to improve oxygen unloading in tissues despite lower arterial oxygen levels.
Increased 2,3-DPG - Adapting to Hypoxia
Increased 2,3-bisphosphoglycerate (2,3-DPG) stabilizes the taut (T) form of hemoglobin, reducing its affinity for oxygen. This promotes O₂ release to tissues, causing a rightward shift in the oxygen-hemoglobin dissociation curve as a key adaptation to chronic hypoxia.
-
Conditions causing ↑ 2,3-DPG:
- High altitude
- Chronic Obstructive Pulmonary Disease (COPD)
- Chronic anemia
- Hyperthyroidism (due to ↑ metabolic rate)
-
Mechanism: Chronic hypoxia enhances anaerobic glycolysis in erythrocytes.
⭐ Stored blood for transfusions is deficient in 2,3-DPG. This leads to ↑ hemoglobin-O₂ affinity, initially impairing oxygen delivery to the recipient's tissues.
📌 Mnemonic: Factors that shift the curve to the Right (↓ affinity): Raised 2,3-DPG, Raised H⁺ (Acidosis), Raised CO₂, and Raised Temperature.
Clinical Correlations - Fetus & Blood Banks
-
Fetal Hemoglobin (HbF)
- Structure: Composed of two α and two γ chains ($α_2γ_2$), which bind 2,3-DPG poorly.
- Result: ↑ Oxygen affinity, causing a left-shift in the oxygen-dissociation curve relative to adult hemoglobin (HbA).
- Function: Enhances O₂ extraction from maternal circulation.
-
Stored Blood for Transfusion
- RBCs lose 2,3-DPG during storage.
- Result: ↑ Hemoglobin affinity for O₂ (left-shift).
- Implication: Transfused blood may initially be impaired in its ability to deliver oxygen to tissues.
⭐ Fetal hemoglobin's higher oxygen affinity is crucial for pulling oxygen across the placental barrier from the mother's lower-affinity adult hemoglobin.
High‑Yield Points - ⚡ Biggest Takeaways
- 2,3-DPG binds to and stabilizes the taut (T) form of deoxyhemoglobin, reducing its oxygen affinity.
- This interaction causes a rightward shift in the oxygen-hemoglobin dissociation curve.
- A rightward shift facilitates oxygen unloading and delivery to peripheral tissues.
- Levels increase during hypoxia and chronic anemia as an adaptive measure.
- Stored blood for transfusions is deficient in 2,3-DPG, impairing O₂ release.
- Fetal hemoglobin (HbF) binds 2,3-DPG poorly, giving it a higher O₂ affinity than adult HbA.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

