Introduction to Fatigue - The Body's Fuse Box
- Definition: A reversible, exercise-induced decline in muscle force-generating capacity. A key protective mechanism, not simple exhaustion.
- Primary Sites:
- Central Fatigue: Originates in the CNS, resulting in ↓ neural drive to the muscle.
- Peripheral Fatigue: Arises from processes at or distal to the neuromuscular junction (NMJ).
- Protective Role: Prevents catastrophic ATP depletion and subsequent irreversible muscle damage (rigor).
⭐ Central fatigue often precedes significant peripheral fatigue, acting as an anticipatory governor to protect muscles from injury.
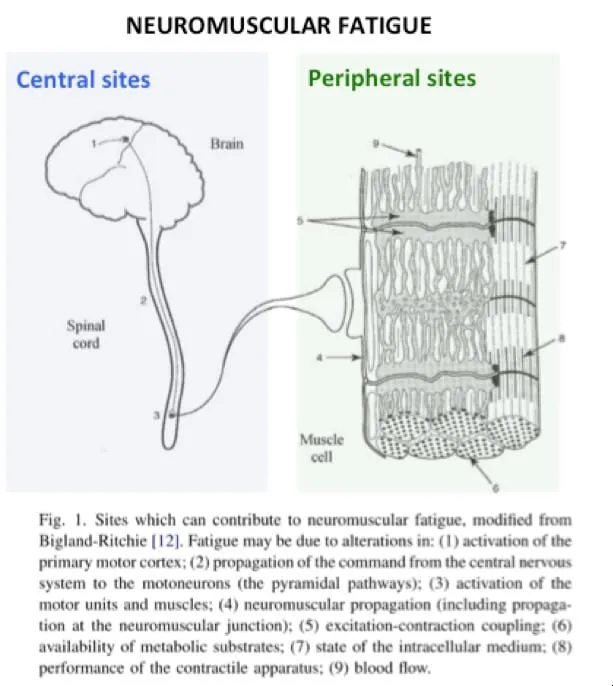
Peripheral Fatigue - Fuel Tank Empty
- Glycogen Depletion: Primary cause of fatigue in prolonged exercise (>60 min). Muscle glycogen is the main fuel for ATP synthesis during intense exercise. Depletion leads to "hitting the wall."
- Phosphocreatine (PCr) Depletion: Occurs rapidly during short, high-intensity bursts (<30s). Limits the rate of ATP regeneration ($PCr + ADP \leftrightarrow ATP + Cr$), reducing power output.
- Hypoglycemia: Depletion of liver glycogen can lead to a fall in blood glucose, impairing CNS function and contributing to fatigue.
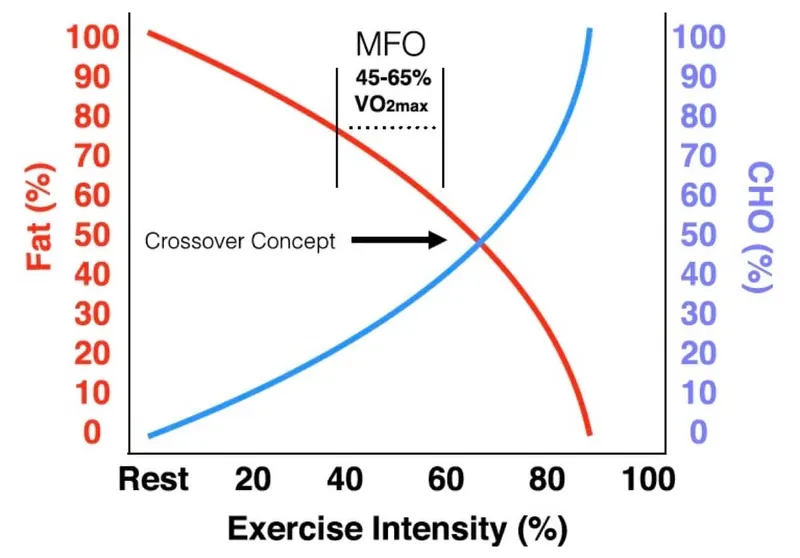
⭐ Exam Favorite: The "crossover concept" dictates that as exercise intensity increases (above ~65% VO₂max), the body shifts from predominantly fat metabolism to carbohydrate metabolism for fuel, accelerating glycogen depletion.
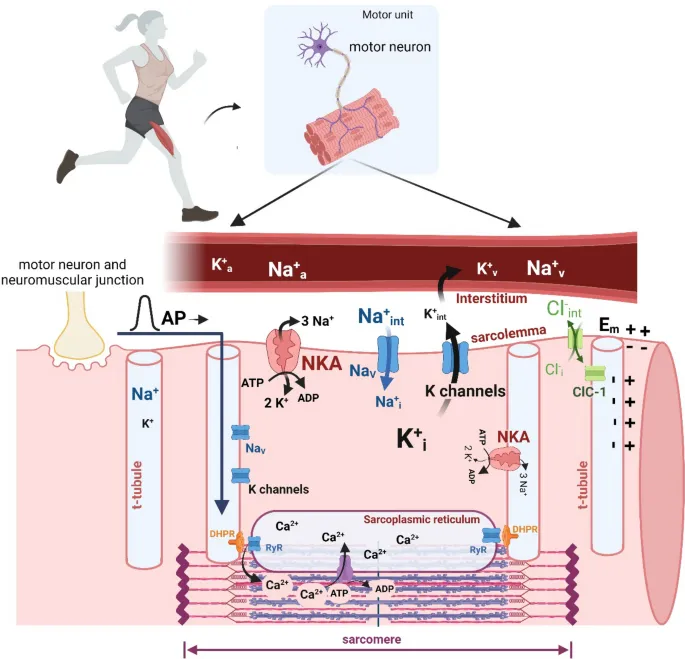
Peripheral Fatigue - Ion Disarray Drama
- Action Potential (AP) Disruption: Intense muscle contraction causes ion shifts, primarily ↑ extracellular K⁺ from repeated repolarization.
- This accumulation alters the resting membrane potential, impairing sarcolemma and T-tubule excitability.
- Result: ↓ AP propagation, leading to ↓ Ca²⁺ release from the sarcoplasmic reticulum (SR).
- Calcium (Ca²⁺) Handling Failure:
- SR Release & Reuptake: Impaired T-tubule APs directly ↓ Ca²⁺ release. SR Ca²⁺-ATPase (SERCA) pump activity also slows.
- Sensitivity: Accumulated H⁺ and inorganic phosphate (Pi) compete with Ca²⁺, reducing troponin's binding sensitivity.
⭐ High-Yield: Elevated extracellular K⁺ can depolarize the muscle fiber membrane sufficiently to inactivate voltage-gated Na⁺ channels, directly hindering the excitability required for sustained contraction.
Central Fatigue - The Brain Says 'Nope'
- CNS-mediated reduction in neural drive to muscles, occurring upstream of the motor neuron, independent of peripheral muscle state.
- Key Mechanisms:
- Neurotransmitter Flux: ↑ Brain Serotonin (5-HT) promotes lethargy. Altered dopamine/norepinephrine signaling reduces motivation and arousal.
- Cerebral Energy Strain: Hypoglycemia can impair brain function and the perceived effort to continue exercise.
- Psychological Override: Perception of effort and motivation can limit performance before true physiological failure.
⭐ The ↑ Serotonin/Dopamine ratio in the brain is strongly linked to feelings of tiredness and reduced motor drive during prolonged exercise.
High‑Yield Points - ⚡ Biggest Takeaways
- Central fatigue involves altered CNS neurotransmission, reducing the neural drive to exercising muscles.
- Peripheral fatigue arises from the motor unit, primarily due to metabolite accumulation and substrate depletion.
- Inorganic phosphate (Pi) accumulation is a key driver, directly impairing cross-bridge cycling and SR Ca²⁺ release.
- Glycogen depletion limits performance in prolonged endurance exercise ("hitting the wall").
- Phosphocreatine (PCr) depletion causes fatigue during short-duration, high-intensity bursts.
- Extracellular K⁺ accumulation can impair sarcolemma excitability.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

