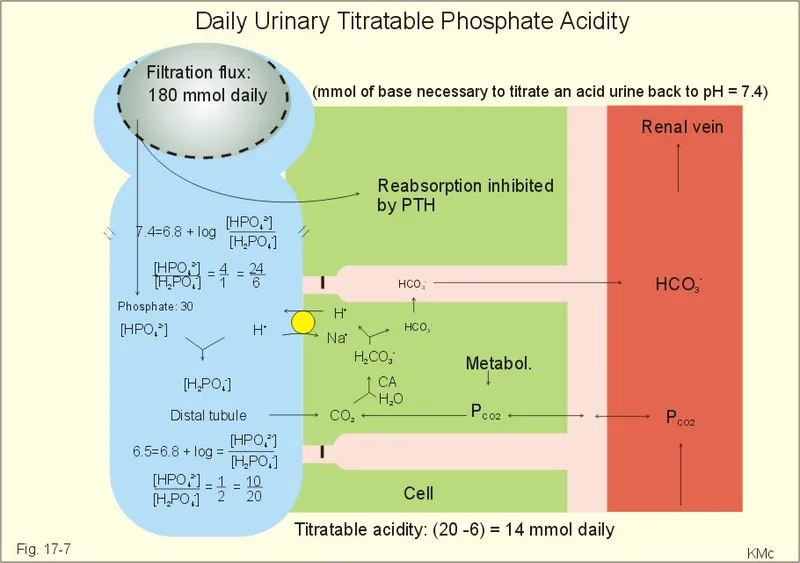
Titratable Acid - The Phosphate Shuttle
- Primary urinary buffer system for excreting non-volatile acids (e.g., from diet).
- Mechanism involves trapping secreted $H^+$ in the tubular fluid by a filtered buffer, primarily phosphate.
- For every $H^+$ secreted and buffered, a new $HCO_3^-$ is generated and returned to the blood, replenishing the body's buffer stores.
- The phosphate buffer system's pKa is ~6.8, making it highly effective in the physiological pH range of urine.
⭐ High-Yield: Titratable acid excretion accounts for eliminating ~30-40 mEq/day of fixed acid. The amount is limited by the quantity of phosphate filtered by the glomerulus.
Regulation & Quantification - Acid Accounting
Titratable Acid (TA) is a measure of protons ($H^+$) excreted in the urine bound to buffers, primarily phosphate ($HPO_4^{2-}$).
- Quantification: Measured by titrating urine with a strong base (e.g., NaOH) back to the normal blood pH of 7.4.
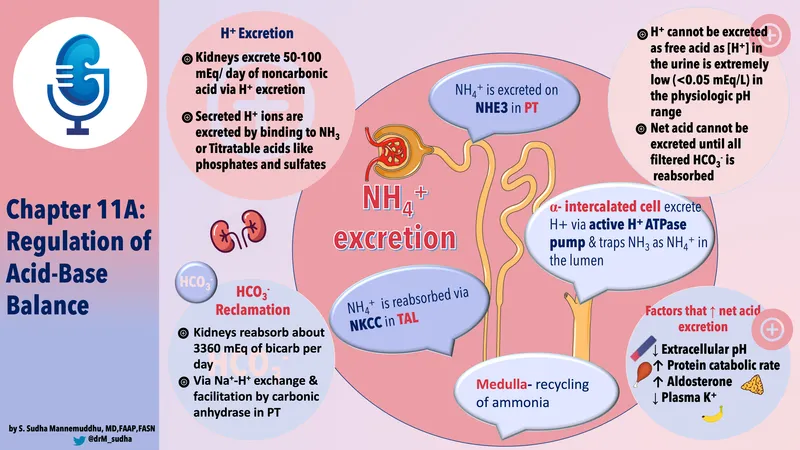
- Net Acid Excretion (NAE): Represents the total renal contribution to acid-base balance.
- $NAE = (\text{Titratable Acid}) + (NH_4^+) - (\text{Urinary } HCO_3^-)$
- Phosphate Buffer System: The main urinary buffer system ($H_2PO_4^- / HPO_4^{2-}$) has a pKa of ~6.8, making it highly effective at physiological urine pH.
- Limitation: TA formation is limited by the quantity of buffers (especially phosphate) filtered by the glomerulus.
⭐ In chronic acidosis, renal ammoniagenesis is the primary, adaptive response for acid excretion, increasing dramatically while titratable acidity remains relatively fixed.
Clinical Context - Acidosis in Action
Metabolic acidosis (e.g., DKA, lactic acidosis) triggers a crucial renal compensatory response to excrete the excess H⁺ load and restore pH.
- Cellular Action: Type A intercalated cells in the collecting ducts ↑ H⁺ secretion into the tubular fluid via apical H⁺-ATPase pumps.
- Urinary Buffering: Secreted H⁺ immediately binds to filtered buffers to prevent a precipitous drop in urine pH.
- Phosphate: The primary titratable acid buffer.
- $H^+ + HPO_4^{2-} \rightarrow H_2PO_4^-$
- This traps H⁺, allowing for its excretion as titratable acid.
⭐ While titratable acid excretion helps, the ammonia buffer system ($NH_3/NH_4^+$) is quantitatively the most important mechanism for excreting large acid loads, especially in chronic acidosis, due to its ability to be significantly upregulated.
High‑Yield Points - ⚡ Biggest Takeaways
- Titratable acid is H⁺ excreted by urinary buffers, mainly the phosphate buffer system (HPO₄²⁻ → H₂PO₄⁻).
- This is key for excreting fixed, non-volatile acids from protein metabolism.
- Occurs in α-intercalated cells of the distal tubule and collecting ducts.
- Accounts for ~1/3 of net acid excretion; ammonium (NH₄⁺) handles the majority.
- Its capacity is limited by the filtered phosphate load, unlike the highly adaptable ammonia system.
- Excretion increases in acidosis but is not significantly upregulated in chronic acidosis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

