Bicarbonate Rescue - The Great Reclaim
- The kidneys reclaim almost all filtered bicarbonate ($HCO_3^-$), with ~85% of this occurring in the Proximal Convoluted Tubule (PCT).
- This is an indirect reabsorption; $HCO_3^-$ itself doesn't cross the apical membrane. The process hinges on proton ($H^+$) secretion.
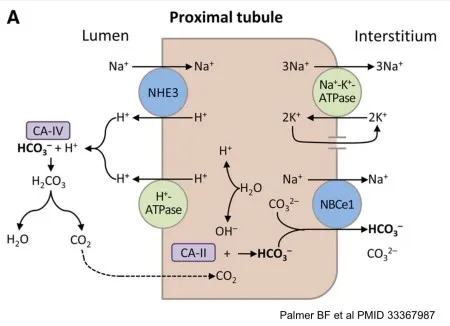
⭐ Carbonic anhydrase inhibitors (e.g., Acetazolamide) block $H^+$ secretion by halting the intracellular production of $H^+$. This prevents $HCO_3^-$ reabsorption, leading to bicarbonaturia and a hyperchloremic metabolic acidosis.
Acid Excretion - Making New Buffer
-
Kidneys generate new $HCO_3^-$ while excreting fixed acids, primarily via two pathways that couple $H^+$ secretion with $HCO_3^-$ synthesis.
-
1. Titratable Acidity:
- Filtered phosphate ($HPO_4^{2-}$) buffers secreted $H^+$ in the tubular fluid.
- $H^+ + HPO_4^{2-} \rightarrow H_2PO_4^-$ (excreted).
- For each $H^+$ secreted, one new $HCO_3^-$ is reabsorbed into the blood.
- Accounts for ~1/3 of new $HCO_3^-$ generation.
-
2. Ammonium ($NH_4^+$) Excretion:
- Proximal tubule cells metabolize glutamine: $Glutamine \rightarrow 2 NH_4^+ + 2 HCO_3^-$.
- The new $HCO_3^-$ enters the blood.
- $NH_4^+$ is secreted into the lumen.
- In the collecting duct, secreted $H^+$ is trapped by diffused ammonia ($NH_3$) forming $NH_4^+$, which is then excreted.
- $NH_3 + H^+ \leftrightarrow NH_4^+$.
⭐ In chronic acidosis, renal glutamine metabolism and $NH_4^+$ excretion increase dramatically, becoming the dominant mechanism for acid excretion.
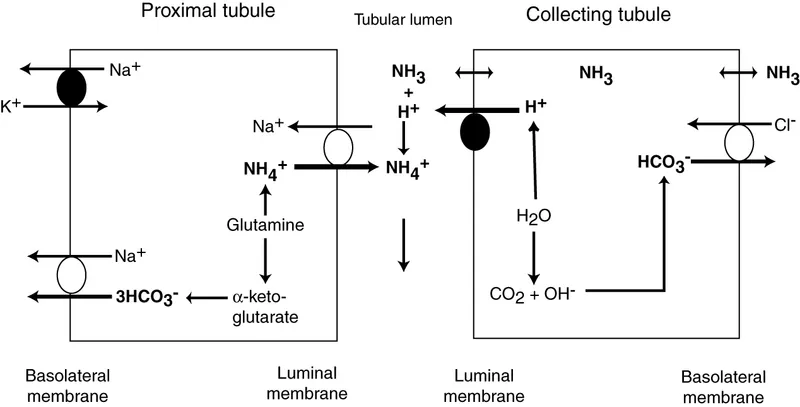
Ammonia Factory - The Glutamine Game
- The kidney's main long-term response to acidosis, primarily occurring in the Proximal Convoluted Tubule (PCT).
- Glutamine is transported into PCT cells and metabolized by glutaminase.
- This process yields two ammonia ($NH_3$) molecules and two new bicarbonate ($HCO_3^−$) molecules.
- $NH_3$ diffuses into the tubular lumen, combining with secreted $H^+$ to form ammonium ($NH_4^+$).
- $NH_4^+$ is charged, trapping it in the tubule for excretion (net acid loss).
- The new $HCO_3^−$ is reabsorbed into the blood, buffering the acidosis.
⭐ In chronic acidosis, renal gene expression for glutaminase is upregulated over 3-5 days, dramatically increasing the kidney's capacity to excrete acid as $NH_4^+$.
Renal Tubular Acidosis - Leaky Pipes
Normal anion gap metabolic acidosis resulting from defective renal acid-base handling. All types cause ↓ serum $HCO_3^-$.
| Type | Defect Location & Mechanism | Serum K⁺ | Urine pH | Key Features |
|---|---|---|---|---|
| Type 1 (Distal) | ↓ H⁺ secretion in distal tubule | ↓ K⁺ | > 5.5 | Chronic kidney stones (calcium phosphate) |
| Type 2 (Proximal) | ↓ $HCO_3^-$ reabsorption in PCT | ↓ K⁺ | < 5.5 | Fanconi syndrome, vitamin D deficiency |
| Type 4 (Hyperkalemic) | Aldosterone deficiency/resistance | ↑ K⁺ | < 5.5 | Common in diabetics; impaired ammoniagenesis |
High‑Yield Points - ⚡ Biggest Takeaways
- The kidneys are the primary long-term regulators of acid-base balance, mainly by reabsorbing filtered HCO₃⁻ and secreting H⁺.
- Bicarbonate reabsorption occurs predominantly in the proximal convoluted tubule (PCT), recovering ~85%.
- Type A intercalated cells in the collecting duct actively secrete H⁺ via an H⁺-ATPase during acidosis.
- Ammoniagenesis in the PCT from glutamine is the principal mechanism for excreting a large acid load.
- Aldosterone stimulates H⁺ secretion, which can contribute to metabolic alkalosis.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

