Metabolic Acidosis - The Acid Test
- Pathophysiology: Primary ↓ in serum bicarbonate ($HCO_3^−$), leading to ↓ blood pH.
- Compensation: Hyperventilation (Kussmaul respirations) to ↓ $pCO_2$.
- Winter's Formula predicts response: Expected $pCO_2 = (1.5 \times [HCO_3^−]) + 8 \pm 2$.
- Anion Gap (AG): Key diagnostic step. $AG = [Na^+] - ([Cl^−] + [HCO_3^−])$. Normal: 8-12 mEq/L.
⭐ Ingestion of ethylene glycol (antifreeze) or methanol are classic causes of high anion gap metabolic acidosis where prompt recognition and treatment are life-saving.
📌 HAGMA Mnemonic (MUDPILES):
- Methanol, Uremia, DKA, Propylene glycol, Iron/Isoniazid, Lactic acidosis, Ethylene glycol, Salicylates.
Anion Gap - Mind the Gap
-
Calculated to differentiate causes of metabolic acidosis.
-
Formula: $AG = Na^+ - (Cl^- + HCO_3^-)$
-
Normal Range: 8-12 mEq/L. Represents unmeasured anions (mostly albumin).
-
High Anion Gap (HAGMA): Caused by accumulation of unmeasured organic acids.
- Glycols, Oxoproline, L-Lactate, D-Lactate, Methanol, Aspirin, Renal failure, Ketoacidosis.
-
Normal Anion Gap (NAGMA): Hyperchloremic acidosis due to loss of bicarbonate.
- Hyperalimentation, Acetazolamide, Renal Tubular Acidosis, Diarrhea, Uretero-enteric fistula, Pancreatic fistula.
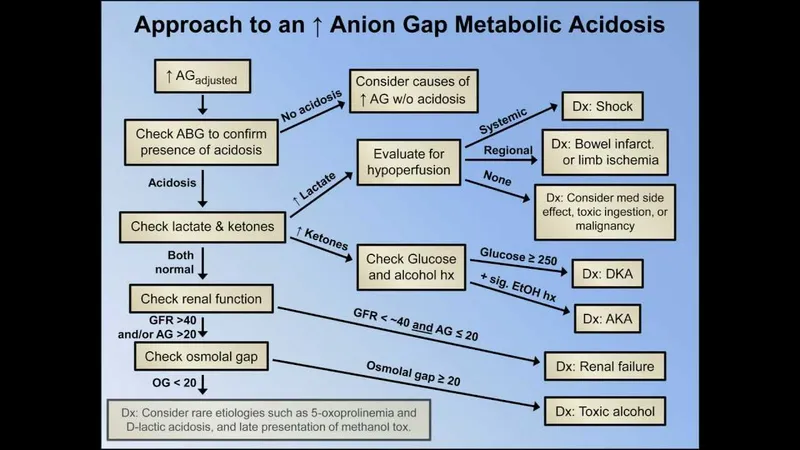
⭐ Always correct the anion gap for albumin. For every 1 g/dL decrease in albumin (from 4.0), add 2.5 to the calculated anion gap.
Normal Anion Gap - No Gap, No Problem?
- Hyperchloremic Metabolic Acidosis: Characterized by loss of bicarbonate ($HCO_3^−$) compensated by an equivalent increase in chloride ($Cl^−$), thus maintaining a normal anion gap.
- Causes (📌 HARDUP):
- Hyperalimentation / Hyperchloremia
- Acetazolamide / Addison's disease
- Renal Tubular Acidosis (RTA)
- Diarrhea (most common cause)
- Ureteroenteric fistula
- Pancreatic fistula
- Key Diagnostic Step: Calculate the Urine Anion Gap (UAG) to differentiate between renal and non-renal causes.
- $UAG = (U_{Na^+} + U_{K^+}) - U_{Cl^−}$
⭐ A negative UAG suggests a non-renal cause like diarrhea. The kidneys are functioning properly and are excreting excess acid as ammonium chloride ($NH_4Cl$), increasing urinary $Cl^−$ and making the UAG negative.
Compensation - The Body's Buffer Zone
- Primary Response: Lungs rapidly expel CO₂ to counteract acidosis.
- Respiratory Compensation: Hyperventilation (Kussmaul respirations) creates a compensatory respiratory alkalosis.
- Predict the expected PaCO₂ with Winter's Formula:
- $Expected PaCO₂ = (1.5 × [HCO₃⁻]) + 8 ± 2$
- If measured PaCO₂ is higher, suspect a concurrent respiratory acidosis.
- Predict the expected PaCO₂ with Winter's Formula:
⭐ Respiratory compensation is fast, initiating within minutes and peaking at 12-24 hours. It is the most important compensatory mechanism in metabolic acidosis.

- Renal Compensation: Slower (days); kidneys increase H⁺ excretion and HCO₃⁻ reabsorption.
High-Yield Points - ⚡ Biggest Takeaways
- Metabolic acidosis is characterized by a primary decrease in serum HCO₃⁻.
- The main compensatory mechanism is hyperventilation to decrease PaCO₂.
- Always calculate the anion gap to narrow the differential diagnosis between HAGMA and NAGMA.
- Use Winter's formula (Expected PaCO₂ = 1.5 × [HCO₃⁻] + 8 ± 2) to determine if respiratory compensation is adequate.
- HAGMA involves the addition of an unmeasured acid (e.g., MUDPILES).
- NAGMA involves the loss of bicarbonate (e.g., HARDASS).
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

