Buffer Basics - Acid-Base Guardians
-
Bicarbonate Buffer System: Primary ECF buffer. Rapidly responds to metabolic acid changes. Governed by the Henderson-Hasselbalch equation:
- $pH = pKa + log([HCO_3^-]/[0.03 * PCO_2])$
- Maintains ECF pH near 7.4.
-
Phosphate Buffer System: Key in intracellular fluid (ICF) and as a urinary buffer.
- Uses the $H_2PO_4^-$/$HPO_4^{2-}$ pair.
- Crucial for excreting H+ in urine.
-
Protein Buffer System: Major ICF and plasma buffer.
- Amphoteric proteins (e.g., albumin) can accept or donate H+.
- Hemoglobin is a vital buffer in RBCs, especially for CO2 transport.
⭐ High-Yield: Hemoglobin is a particularly effective buffer due to its high concentration in blood and the numerous histidine residues, which have a pKa near physiological pH.
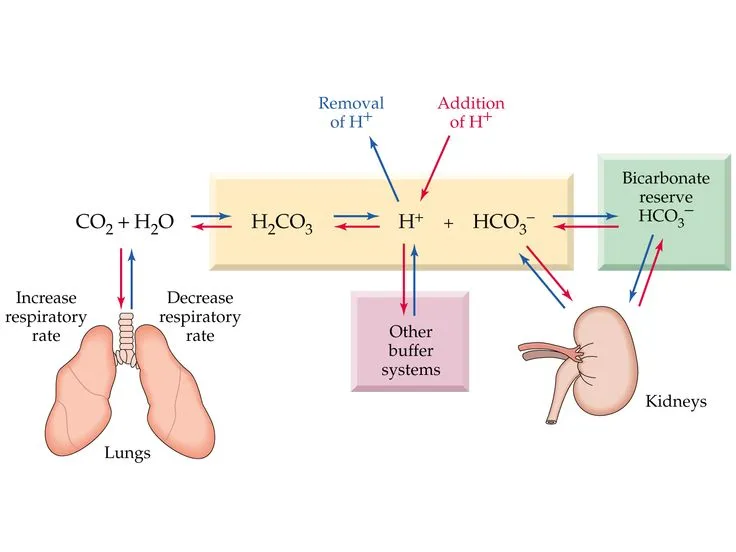
Bicarbonate System - The Lung-Kidney Tango
- The most critical buffer in the extracellular fluid (ECF), responsible for managing moment-to-moment pH changes.
- Equation: $CO_2 + H_2O \leftrightarrow H_2CO_3 \leftrightarrow H^+ + HCO_3^-$
- Lungs (Fast Control): Regulate volatile acid by adjusting $PCO_2$ via ventilation.
- ↑Ventilation → Blow off $CO_2$ → ↓$PCO_2$ → ↑pH
- Kidneys (Slow Control): Regulate fixed acids by adjusting $[HCO_3^-]$ reabsorption and generation.
- Slower, more powerful response (hours to days).
⭐ The system's pKa is 6.1, which is not ideal for buffering at physiological pH (7.4). Its effectiveness stems from the independent and powerful regulation of $CO_2$ by the lungs and $HCO_3^-$ by the kidneys.
Phosphate & Proteins - The Supporting Cast
-
Phosphate Buffer System: A key player in intracellular fluid (ICF) and renal tubules.
- Components: Dihydrogen phosphate ($H_2PO_4^−$, the weak acid) and monohydrogen phosphate ($HPO_4^{2−}$, the conjugate base).
- Reaction: $H_2PO_4^− \leftrightarrow H^+ + HPO_4^{2−}$
- Has an effective pKa of 6.8, making it an efficient buffer in urine and ICF.
-
Protein Buffer System: The most abundant buffer system, particularly intracellularly.
- Hemoglobin (Hb) in RBCs is the most significant protein buffer.
- Proteins are amphoteric: carboxyl groups (-COOH) donate $H^+$, while amino groups (-NH₂) accept $H^+$.
- The amino acid Histidine is the most effective amino acid buffer.
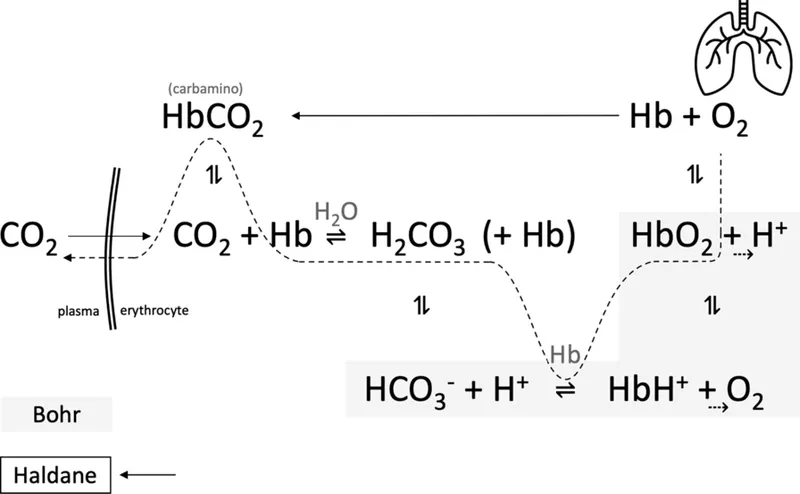
⭐ High-Yield: The Haldane Effect is critical for $CO_2$ transport. Deoxygenated hemoglobin in the tissues is a better proton acceptor (stronger base) than oxygenated Hb. It readily binds to $H^+$ ions formed when $CO_2$ is converted to bicarbonate, thus facilitating the loading and transport of $CO_2$ from tissues to the lungs.
High‑Yield Points - ⚡ Biggest Takeaways
- The bicarbonate buffer system is the primary ECF buffer, dynamically regulated by the lungs (CO2) and kidneys (HCO3⁻).
- Proteins are the most plentiful buffers, with hemoglobin being crucial in RBCs and albumin in plasma.
- The phosphate buffer system is most effective in the ICF and renal tubules.
- The Henderson-Hasselbalch equation is fundamental for understanding the bicarbonate system's function.
- All buffers are linked by the isohydric principle, maintaining a common pH.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

