HIV Entry - Blocking the Gates
- CCR5 Antagonist: Maraviroc
- Allosterically binds to the host cell CCR5 coreceptor, preventing interaction with viral gp120.
- 📌 Maraviroc stops HIV from marrying the T-cell at the CCR5 door.
- Fusion Inhibitor: Enfuvirtide
- Binds to the first heptad repeat (HR1) of the viral gp41 subunit, preventing the conformational change required for membrane fusion.
- Administered via subcutaneous injection; risk of local injection site reactions.
⭐ Tropism Assay Required: Before initiating Maraviroc, an assay must confirm the virus is exclusively CCR5-tropic. It is not active against CXCR4-tropic or dual/mixed-tropic HIV-1.
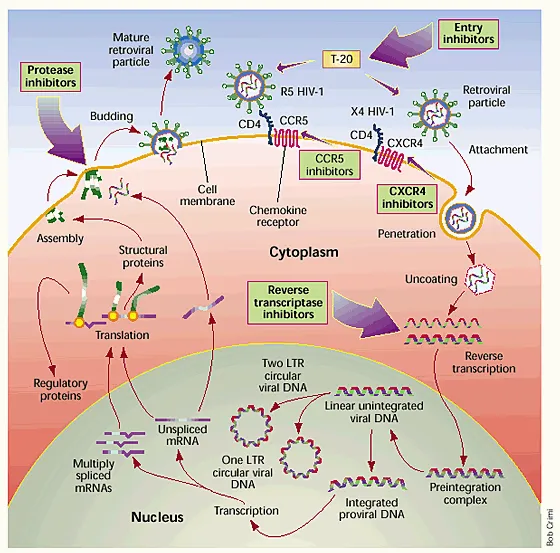
Entry Inhibitors - The Agents
-
CCR5 Antagonist: Maraviroc
- Mechanism: Binds to the human CCR5 co-receptor, preventing viral gp120 from interacting with it. This blocks entry of CCR5-tropic HIV strains.
- Metabolism: Substrate of CYP3A4; dose adjustments needed with inhibitors/inducers.
- Adverse Effects: Hepatotoxicity (may be preceded by a systemic allergic reaction), postural hypotension.
- 📌 Mnemonic: Maraviroc blocks the CCR5 rock (receptor).
-
Fusion Inhibitor: Enfuvirtide (T-20)
- Mechanism: A synthetic peptide that binds to the viral gp41 subunit, preventing the conformational change required for membrane fusion.
- Use: Reserved for treatment-experienced patients with multidrug-resistant HIV.
- Administration: Subcutaneous injection twice daily.
- Adverse Effects: Almost universal injection-site reactions (pain, erythema, nodules); increased risk of bacterial pneumonia.
-
Attachment Inhibitor: Fostemsavir
- Mechanism: A prodrug converted to temsavir, which binds directly to viral gp120, inhibiting attachment to host CD4 cells.
⭐ Exam Favorite: Before starting Maraviroc, a tropism assay (e.g., Trofile) is mandatory to confirm the patient has a CCR5-tropic virus. It is ineffective against CXCR4-tropic or dual/mixed-tropic HIV variants.
Clinical Use & Resistance - Salvage Strategy
- Primary Role: Salvage therapy for treatment-experienced patients with multi-drug resistant (MDR) HIV-1. Not for initial therapy.
- Maraviroc (CCR5 Antagonist):
- Only for CCR5-tropic virus (requires tropism test).
- Resistance: Shift to CXCR4-tropism or gp120 mutations.
- Enfuvirtide (Fusion Inhibitor):
- For MDR-HIV with limited options due to injection site reactions and cost.
- Resistance: Mutations in gp41.
- Ibalizumab & Fostemsavir:
- Newer options for heavily treatment-experienced adults with MDR-HIV.
⭐ Before starting Maraviroc, a tropism assay is mandatory. It is only effective against CCR5-tropic HIV, not CXCR4-tropic or dual-tropic strains.
Adverse Effects - The Trade-Offs
-
Maraviroc (CCR5 Antagonist):
- ⚠️ Black Box Warning: Hepatotoxicity, often preceded by a systemic allergic reaction (e.g., rash, eosinophilia).
- Cardiovascular events (e.g., orthostatic hypotension).
- Increased risk of upper respiratory infections.
-
Enfuvirtide (Fusion Inhibitor):
- Local injection site reactions are nearly universal (>90%), causing pain, erythema, and nodules.
- Increased risk of bacterial pneumonia.
- Systemic hypersensitivity reactions.
⭐ Before starting Maraviroc, a tropism test is mandatory to confirm CCR5-tropic HIV-1 strain.
High‑Yield Points - ⚡ Biggest Takeaways
- Maraviroc binds the host CCR5 coreceptor, blocking interaction with viral gp120. A tropism assay is required.
- Enfuvirtide binds viral gp41, inhibiting the conformational change for membrane fusion.
- Ibalizumab is a monoclonal antibody against CD4, acting as a post-attachment inhibitor.
- These agents are typically reserved for salvage therapy in multidrug-resistant HIV.
- Key toxicities: hepatotoxicity for Maraviroc and severe injection-site reactions for Enfuvirtide.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

