HBV Lifecycle & Targets - Viral Blueprint
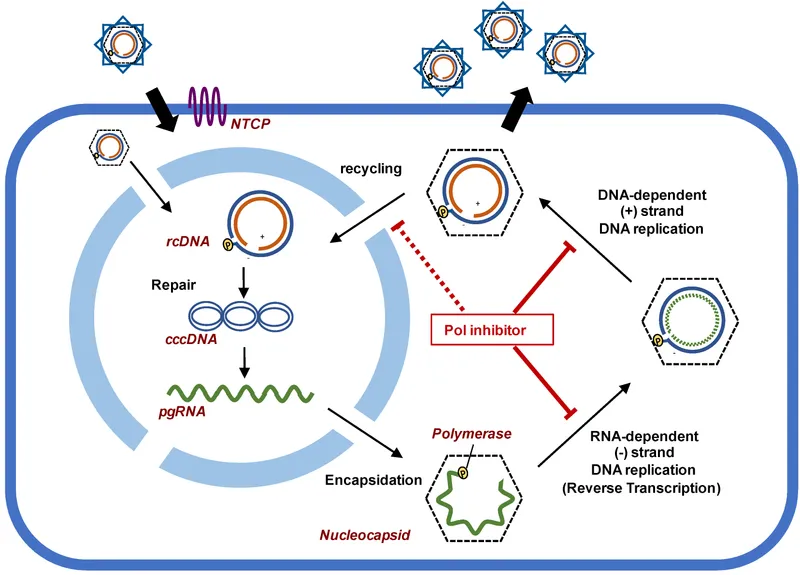
- Core Process: A DNA virus that uniquely replicates via an RNA intermediate using reverse transcriptase.
- Viral Reservoir: Forms a stable, episomal covalently closed circular DNA ($cccDNA$) in the hepatocyte nucleus.
- $cccDNA$ serves as a persistent template for all viral gene products.
- Not cleared by current therapies, leading to lifelong infection.
- Primary Drug Targets:
- Reverse Transcriptase (RT): Targeted by Nucleos(t)ide Reverse Transcriptase Inhibitors (NRTIs) like Tenofovir & Entecavir.
- Entry: Blocked by Bulevirtide.
⭐ The persistence of nuclear cccDNA is the primary reason for treatment failure and viral rebound after discontinuing NRTIs. It's the key barrier to a sterilizing cure.
Nucleos(t)ide Analogs - The Chain Breakers
-
Mechanism of Action: Competitively inhibit HBV DNA polymerase/reverse transcriptase. After incorporation into the growing viral DNA strand, they cause premature chain termination, halting viral replication.
-
First-Line Agents:
- Tenofovir (TDF or TAF): Potent with a high barrier to resistance.
- TDF: Associated with nephrotoxicity (e.g., Fanconi syndrome) and decreased bone mineral density.
- TAF (alafenamide): Prodrug with lower plasma levels, resulting in fewer renal and bone side effects.
- Entecavir: Potent, high barrier to resistance. Must be taken on an empty stomach.
- Tenofovir (TDF or TAF): Potent with a high barrier to resistance.
-
Other Agents:
- Lamivudine, Telbivudine: Lower barrier to resistance.
- Adefovir: Risk of dose-dependent nephrotoxicity.
-
Class-wide Warnings:
- Rare but severe risk of lactic acidosis and hepatomegaly with steatosis.
⭐ Exam Favorite: Abrupt discontinuation of these agents can cause a severe acute exacerbation of Hepatitis B, potentially leading to hepatic failure. Always taper or switch therapy under guidance.
Interferon-Alpha - The Immune Alarm
- Mechanism: A host cytokine that boosts anti-viral immune responses. It doesn't target HBV directly but activates host defenses, like setting off an "immune alarm."
- Induces expression of MHC class I molecules, enhancing cytotoxic T-cell recognition of infected hepatocytes.
- Activates natural killer (NK) cells.
- Clinical Use: Finite course of therapy (48 weeks). Often considered in younger patients with well-compensated liver disease and high serum ALT.
- Adverse Effects:
- Flu-like symptoms (fever, chills, myalgia) are very common.
- Neuropsychiatric: Depression, anxiety, irritability.
- Bone marrow suppression.
⭐ Pegylated interferon (Peg-IFN) is conjugated with polyethylene glycol, which ↑ half-life, allowing for once-weekly injections and providing a more sustained therapeutic effect.
- Goal of therapy is HBV DNA suppression, not a cure; lifelong treatment is common.
- First-line agents are Entecavir and Tenofovir (TDF/TAF) due to high potency and a high barrier to resistance.
- All are NRTIs that inhibit HBV DNA polymerase.
- Tenofovir Disoproxil Fumarate (TDF) is associated with nephrotoxicity and decreased bone mineral density.
- Tenofovir Alafenamide (TAF) is a newer prodrug with a better renal and bone safety profile.
- Pegylated interferon-alfa offers a finite treatment course but has significant side effects.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

