CMV Antivirals - The First Responders
-
Ganciclovir & Valganciclovir (oral prodrug)
- MOA: Guanosine analog. Must be phosphorylated by viral kinase UL97 to inhibit CMV DNA polymerase.
- Toxicity: Myelosuppression (neutropenia, thrombocytopenia), fever, rash.
-
Foscarnet
- MOA: Pyrophosphate analog. Directly inhibits DNA polymerase. No viral kinase activation needed.
- Toxicity: Nephrotoxicity, electrolyte imbalances (↓Ca²⁺, ↓Mg²⁺, ↓K⁺).
- 📌 Mnemonic: Foscarnet chelates fosphate & other minerals.
-
Cidofovir
- MOA: Nucleotide analog. Bypasses need for viral kinase.
- Toxicity: Severe dose-dependent nephrotoxicity. Administer with probenecid.
⭐ Resistance: Mutations in the UL97 gene prevent ganciclovir phosphorylation, conferring resistance. Foscarnet or cidofovir are used as alternatives.
Resistance & Rescue - The Backup Crew
-
Primary Driver: Mutations in viral genes, most commonly selected for under drug pressure in immunocompromised hosts.
-
Mechanisms of Resistance:
- Ganciclovir/Valganciclovir: Most common resistance via UL97 gene mutation (a viral phosphotransferase). This prevents the crucial first monophosphorylation step needed for drug activation.
- Foscarnet/Cidofovir: Resistance via mutations in the UL54 gene, which codes for the viral DNA polymerase itself. Can confer cross-resistance.
⭐ The most frequent cause of ganciclovir resistance is a mutation in the UL97 gene, which blocks the initial, necessary phosphorylation step for drug activation.
- Management of Resistance:
- 📌 Foscarnet Functions on the polymerase directly; Ganciclovir Gets stopped by UL97 mutations.
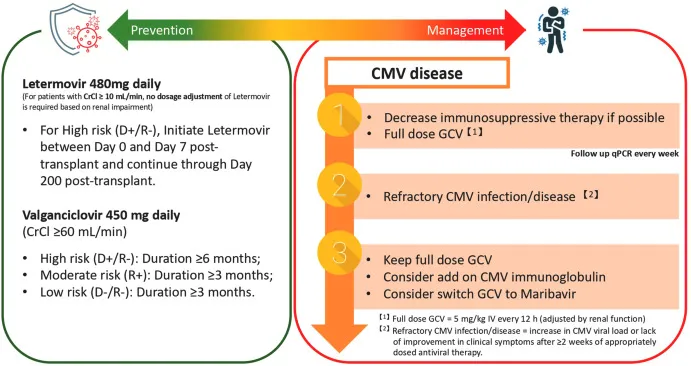
Prophylaxis Power - The Preventative Play
- Goal: Prevent CMV end-organ disease (retinitis, colitis, pneumonitis) in high-risk, immunocompromised hosts.
- High-Risk Scenarios:
- Solid Organ Transplant (SOT): Highest risk in CMV D+/R- (Donor positive/Recipient negative).
- Allogeneic Hematopoietic Stem Cell Transplant (HSCT).
- First-Line Prophylaxis: Valganciclovir (oral ganciclovir prodrug). Requires renal dose adjustments and monitoring for myelosuppression.
- HSCT-Specific Prophylaxis: Letermovir.
- Novel mechanism: Inhibits the CMV terminase complex, crucial for viral DNA processing.
- 📌 Letermovir lets the marrow live (no myelosuppression) & terminates CMV.
⭐ Letermovir is used for CMV-seropositive [R+] allogeneic HSCT recipients. Prophylaxis typically starts post-engraftment and continues through day +100 post-transplant.
High‑Yield Points - ⚡ Biggest Takeaways
- Ganciclovir and its prodrug valganciclovir are first-line for CMV, but cause significant myelosuppression.
- Ganciclovir requires viral kinase UL97 for activation; mutations in this gene confer resistance.
- Foscarnet and cidofovir treat ganciclovir-resistant CMV as they do not require viral kinase activation.
- Foscarnet is nephrotoxic and causes significant electrolyte imbalances (Ca²⁺, PO₄³⁻, Mg²⁺).
- Cidofovir's nephrotoxicity is managed with probenecid and saline hydration.
- Letermovir is for prophylaxis in HSCT patients, inhibiting the CMV terminase complex.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

