Gene Therapy 101 - Fixing Faulty Blueprints
- Core Concept: A technique to treat or cure genetic disorders by modifying a person's genes.
- Key Strategies:
- Gene Replacement: A functional gene is introduced to replace a mutated one.
- Gene Inactivation: The mutated gene is 'switched off'.
- Gene Editing: Correcting the DNA sequence directly (e.g., CRISPR-Cas9).
- Vectors: Modified viruses (Adeno-Associated Virus - AAV, Lentivirus) are the most common delivery vehicles.
⭐ Zolgensma (onasemnogene abeparvovec) is a landmark AAV-based gene therapy for Spinal Muscular Atrophy (SMA), replacing the defective SMN1 gene.
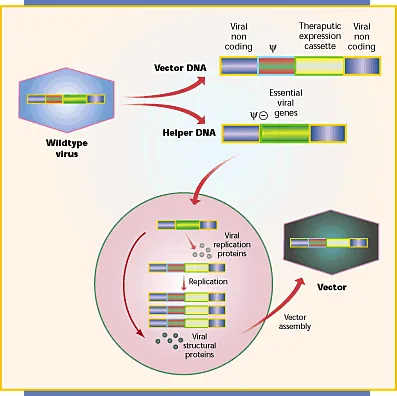
Genetic Mail Carriers - Viral & Non-Viral Vectors
Gene therapy relies on vectors to deliver therapeutic genes into target cells. The choice of vector is critical and depends on the specific application, balancing efficiency against safety.
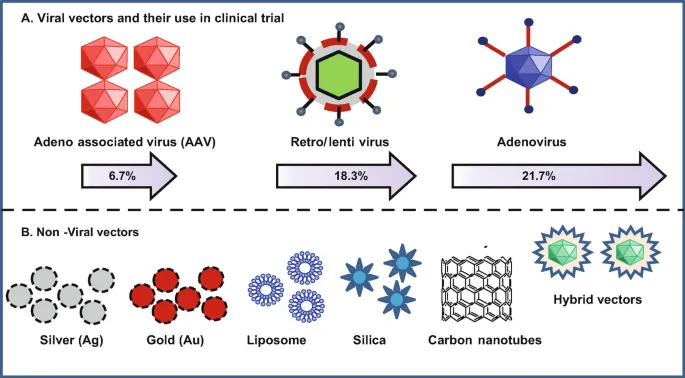
| Feature | Viral Vectors | Non-Viral Vectors |
|---|---|---|
| Mechanism | Use natural viral ability to infect cells | Chemical or physical methods |
| Examples | Adeno-associated virus (AAV), Lentivirus, Adenovirus | Liposomes, Nanoparticles, Electroporation |
| Efficiency | High transduction efficiency | Low to moderate efficiency |
| Safety Profile | Risk of immunogenicity, insertional mutagenesis | Safer, lower immunogenicity, non-integrating |
| Cargo Size | Limited capacity | Larger gene capacity |
📌 Mnemonic: "All Little Angels" for common viral vectors: AAV, Lentivirus, Adenovirus.
New Cures on Block - Landmark Treatments
-
Zolgensma (Onasemnogene abeparvovec):
- For Spinal Muscular Atrophy (SMA) Type 1.
- In-vivo therapy using an AAV9 vector to deliver a functional SMN1 gene.
- Notably crosses the blood-brain barrier.
-
Luxturna (Voretigene neparvovec):
- For Leber's Congenital Amaurosis (LCA) from biallelic RPE65 mutations.
- In-vivo therapy delivered via subretinal injection.
-
Libmeldy (Atidarsagene autotemcel):
- For early-onset Metachromatic Leukodystrophy (MLD).
- Ex-vivo therapy using a lentiviral vector to insert a functional ARSA gene into patient hematopoietic stem cells (HSCs).
-
Casgevy (Exagamglogene autotemcel):
- For Sickle Cell Disease & β-thalassemia.
- First approved CRISPR/Cas9-based therapy.
- Ex-vivo editing of HSCs to increase fetal hemoglobin (HbF).
⭐ Zolgensma is famed as one of the world's most expensive drugs, with a single dose costing over $2 million. Its one-time administration offers a potential cure for SMA type 1.
CRISPR & Challenges - Editing The Future
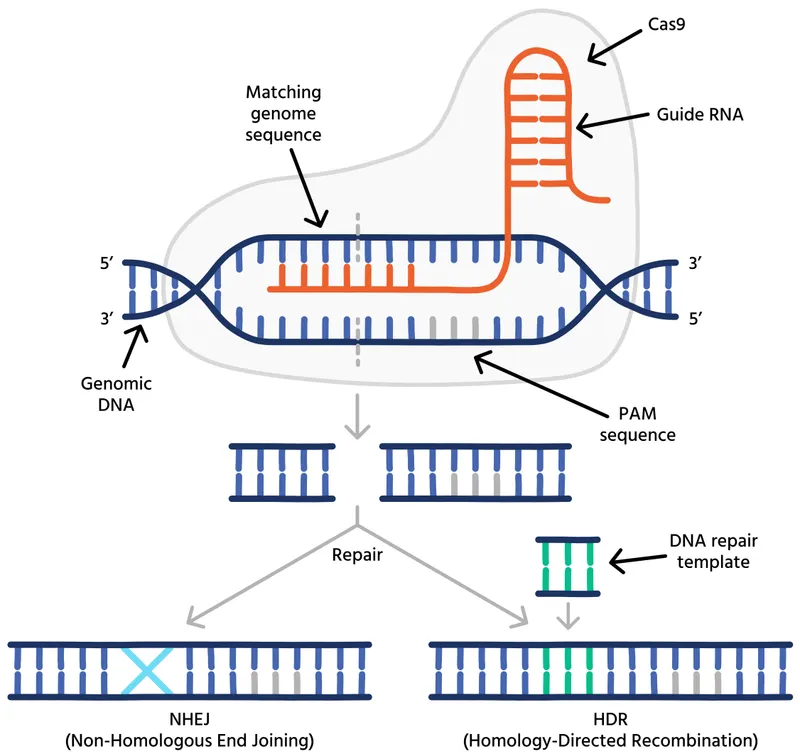
- CRISPR-Cas9: A revolutionary gene-editing tool. Comprises Cas9 nuclease ("molecular scissors") and a guide RNA (gRNA) to target specific DNA sequences.
- Mechanism: Creates double-strand breaks (DSBs) at the target site.
- NHEJ (Non-Homologous End Joining): Inactivates genes (knockout).
- HDR (Homology Directed Repair): Corrects mutations (knock-in).
⭐ High-Yield: Exagamglogene autotemcel (Exa-cel/Casgevy) is the first FDA-approved CRISPR therapy. It targets the BCL11A gene to treat Sickle Cell Disease & β-thalassemia by increasing fetal hemoglobin (HbF) production.
- Challenges:
- Off-target effects: Unintended DNA cuts.
- Delivery: Efficient in vivo delivery to target cells remains a hurdle.
- Ethical concerns: Particularly regarding germline editing.
High‑Yield Points - ⚡ Biggest Takeaways
- Zolgensma (onasemnogene abeparvovec) for Spinal Muscular Atrophy (SMA) uses an AAV9 vector to deliver a functional SMN1 gene.
- Luxturna (voretigene neparvovec) is the approved therapy for RPE65 mutation-associated retinal dystrophy.
- CAR-T cell therapy (e.g., Kymriah) is a revolutionary treatment for relapsed/refractory B-cell ALL.
- Gene therapies for hemoglobinopathies like Thalassemia and Sickle Cell Disease are a key area of research.
- Distinguish ex-vivo (CAR-T) from in-vivo (Zolgensma) gene delivery methods.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

