Cellular Senescence - The Ticking Clock
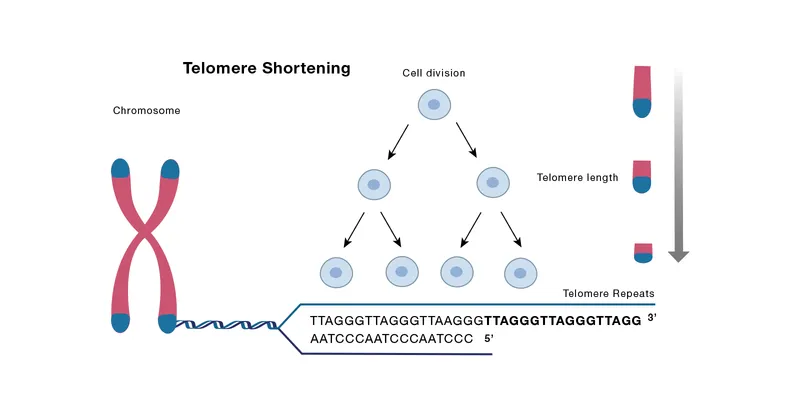
- Telomere Attrition: Finite cell divisions cause progressive shortening of chromosome ends (telomeres), acting as a mitotic clock.
- Hayflick Limit: Normal somatic cells arrest after a fixed number of divisions (~50-70), entering senescence.
- Mechanism: Critically short telomeres are sensed as DNA damage, activating tumor suppressor pathways (p53, Rb) to halt the cell cycle.
⭐ Senescent cells are not inert; they adopt a Senescence-Associated Secretory Phenotype (SASP), releasing pro-inflammatory cytokines (IL-6, IL-8) that can promote chronic inflammation and aging-related diseases.
Telomere Shortening - The End-Replication Problem
- Telomeres: Repetitive DNA sequences (TTAGGG) capping chromosome ends to prevent degradation and fusion.
- End-Replication Problem: DNA polymerase cannot fully replicate the 3' end of the lagging strand, causing progressive telomere shortening with each cell division.
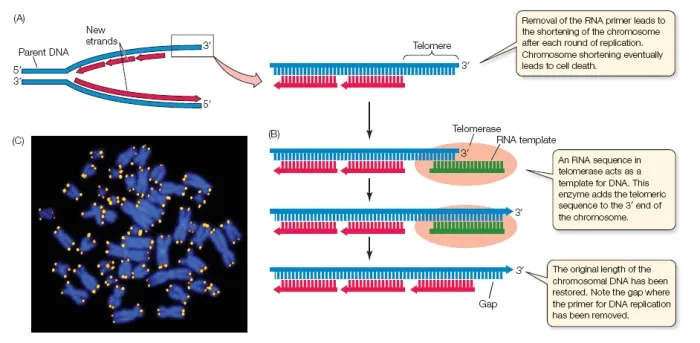
- Hayflick Limit: The finite number of divisions a normal cell can undergo before shortening triggers senescence.
- Telomerase: A reverse transcriptase that adds telomeric DNA.
- Active in germ cells & stem cells, maintaining their length.
- Inactive in most somatic cells.
⭐ Reactivation of telomerase is a hallmark of ~90% of cancers, granting cells replicative immortality by overcoming the Hayflick limit.
Pathways & Proteins - The Cellular Housekeeping
- Proteostasis Collapse: Age-related decline in maintaining a healthy cellular proteome, leading to the accumulation of misfolded or damaged proteins.
- Key Pathways & Their Decline:
- Chaperone-mediated folding: ↓ Heat Shock Proteins (HSPs) impair correct protein folding.
- Ubiquitin-Proteasome System (UPS): Reduced efficiency in tagging (ubiquitination) and degrading cytosolic proteins.
- Autophagy-Lysosome Pathway: Impaired "self-eating" mechanism for clearing damaged organelles and long-lived proteins.
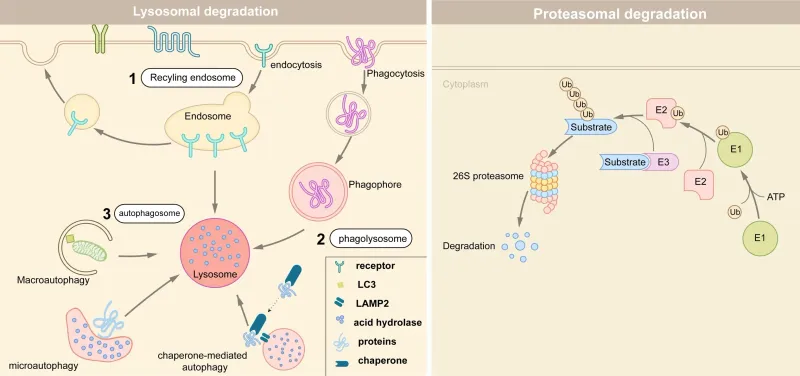
⭐ Lipofuscin: The yellow-brown "wear-and-tear" pigment seen in aging cells (heart, liver, neurons) is composed of oxidized lipid and protein aggregates within lysosomes, representing evidence of cumulative oxidative damage and declining autophagic clearance.
- Telomere attrition is a key driver of replicative senescence, limiting cell divisions.
- Accumulation of DNA damage and mutations, often from reactive oxygen species (ROS), contributes significantly.
- Failed proteostasis leads to the buildup of misfolded proteins, impairing cellular function.
- Deregulated nutrient sensing, particularly involving the mTOR pathway, influences aging.
- Mitochondrial dysfunction results in decreased ATP production and increased ROS, creating a vicious cycle.
- Stem cell exhaustion depletes the regenerative potential of tissues over time.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

