Epidemiology & Risks - The Double Trouble
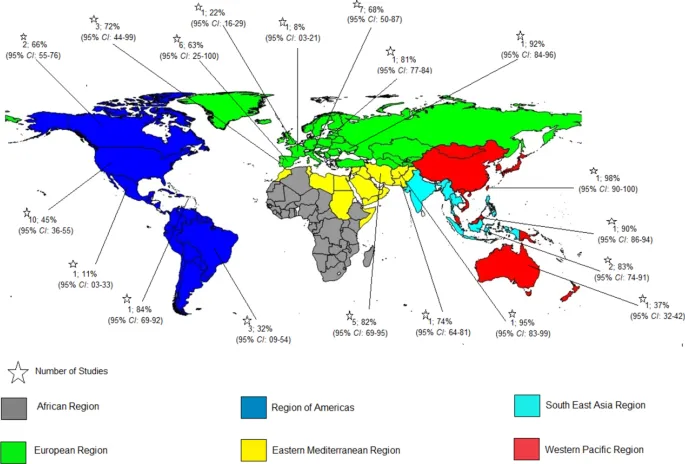
- Shared Transmission: Primarily via parenteral routes.
- Intravenous drug use (IVDU) is the leading risk factor.
- Other risks: high-risk sexual contact, vertical transmission, unsafe medical procedures.
- Accelerated Disease: Co-infection significantly ↑ risk and speed of progression to cirrhosis, hepatic decompensation, and hepatocellular carcinoma (HCC) compared to monoinfection.
⭐ In many co-infected individuals, HBV is dominant, leading to suppressed HCV replication (low HCV RNA). HCV can flare after HBV treatment.
Pathophysiology - Viral Tug-of-War
- Viral Interference: HBV & HCV are hepatotropic viruses that compete for host resources, leading to a pattern of viral dominance where one virus actively suppresses the replication of the other.
- Common Scenario: HCV usually dominates, leading to:
- ↓ HBV DNA levels (often to undetectable levels).
- A state of "occult HBV infection" (HBsAg-negative, but anti-HBc positive).
- Clinical Significance: The viral balance is dynamic. The suppressed virus can reactivate if the dominant one is cleared, e.g., during antiviral therapy.
⭐ Reactivation of HBV upon treatment of HCV with direct-acting antivirals (DAAs) is a major clinical concern, potentially causing fulminant hepatitis. Always screen for HBV (HBsAg, anti-HBs, anti-HBc) before initiating HCV therapy.
Diagnosis & Serology - The Viral Fingerprints
- Initial Dual Screening: Simultaneously test for HBsAg and anti-HCV antibodies in all at-risk patients.
- Confirmation with NAT: Positive screens require Nucleic Acid Testing (NAT) for active infection confirmation.
- HBV DNA (quantitative)
- HCV RNA (quantitative)
- HBV Panel: A full HBV panel (HBsAg, anti-HBs, anti-HBc) is crucial to determine immune status or chronic infection stage.
- Occult HBV Infection (OBI): Common scenario where HBsAg is negative, but anti-HBc is positive. Detectable low-level HBV DNA confirms OBI.
⭐ High-Yield: HCV is the dominant virus; it often suppresses HBV replication. Treating HCV with Direct-Acting Antivirals (DAAs) can lead to a dangerous flare of previously latent HBV.
Treatment Algorithms - The Suppression Strategy
- Primary Goal: Achieve HCV Sustained Virologic Response (SVR) while preventing HBV reactivation.
- Core Principle: Initiate HCV treatment with Direct-Acting Antivirals (DAAs) first.
- Mandatory Monitoring: All patients must be monitored for HBV reactivation before and during HCV therapy.
⭐ FDA Black Box Warning: Treating HCV with DAAs carries a risk of HBV reactivation in co-infected patients. This can lead to fulminant hepatitis, hepatic failure, or death. Screen all patients for HBV before starting DAAs.
Complications & Prognosis - The Long Game
- Accelerated Fibrosis: Faster progression to cirrhosis, decompensated liver disease (ascites, encephalopathy), and hepatocellular carcinoma (HCC) compared to mono-infection.
- ↑ Mortality: Significantly higher risk of liver-related death.
- Viral Interaction: HCV is often dominant, leading to suppression of HBV replication (↓ HBV DNA).
⭐ Reactivation Risk: Treating HCV with Direct-Acting Antivirals (DAAs) can cause a flare of HBV replication, potentially leading to fulminant hepatitis. Screen for HBV before starting HCV therapy.
High‑Yield Points - ⚡ Biggest Takeaways
- Co-infection with HBV and HCV dramatically accelerates progression to severe liver disease, including cirrhosis and hepatocellular carcinoma (HCC).
- Viral interference is common, with HCV often suppressing HBV replication, leading to low or undetectable HBV DNA.
- Crucially, screen for HBV (HBsAg, anti-HBc) before starting HCV direct-acting antivirals (DAAs) to prevent HBV reactivation.
- HBV reactivation during or after DAA therapy can cause fulminant hepatitis.
- Management is complex, often requiring dual therapy and specialist consultation.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

