N-of-1 Trials - The Ultimate Personalization
- Concept: A single-patient, multi-period crossover trial where the patient serves as their own control.
- Goal: To determine the optimal treatment for an individual, not to generalize to a population.
- Design Features:
- Randomized order of treatment and placebo/alternative therapy.
- Washout periods are critical to prevent carryover effects.
⭐ High-Yield: Best suited for chronic, stable conditions with treatments that have a rapid onset and short duration of action (e.g., managing chronic pain, ADHD, or hypertension).
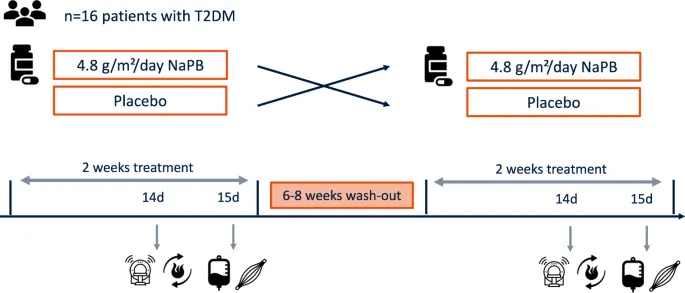
Trial Blueprint - Designing for One
- An N-of-1 trial is a multi-period crossover study within a single patient, who serves as their own control to determine optimal therapy.
- Core design components:
- Randomization: The sequence of treatments (e.g., active drug vs. placebo) is randomly assigned to prevent bias from time or ordering effects.
- Washout Periods: Critical intervals between treatment periods to allow the effects of the previous intervention to dissipate, preventing carryover.
- Blinding: Ideally, both the patient and the clinician are blinded to the treatment being administered in any given period.
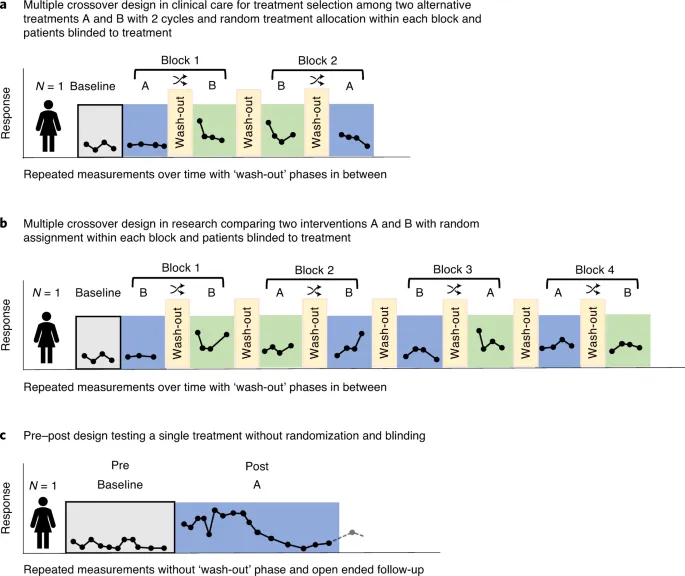
⭐ N-of-1 trials provide the highest level of evidence for a treatment decision in a single patient, representing a cornerstone of personalized medicine.
Weighing the Evidence - Solo Act Strengths
- Ultimate Personalization: Provides the highest level of evidence for treatment effectiveness in a single patient. It directly answers the question: "Does this treatment work for me?"
- Strong Causal Inference: By using multiple crossover periods with treatment and placebo/alternative, it establishes a clearer cause-and-effect relationship for that individual, minimizing confounding.
- Ideal for Chronic, Stable Conditions: Best suited for diseases where the outcome is measurable and the condition is relatively stable (e.g., chronic pain, hypertension, asthma).
- Reduces Bias: Can incorporate key features of larger trials like randomization of treatment periods and double-blinding, enhancing validity.
⭐ Highest Evidence for Individual Care: An N-of-1 trial is considered Level 1 evidence for making a therapeutic decision for a specific patient, surpassing even large RCTs which provide average population effects.
Analysis & Interpretation - Crunching the Numbers
- Primary Analysis: Focuses on the individual, not group averages.
- Visual Analysis: The initial step. Plotting outcome data over time (run charts) to visually inspect for trends, cycles, and treatment effects.
- Statistical Methods:
- Paired t-test: Commonly used to compare the mean outcome between pairs of treatment periods (e.g., A vs. B).
- Bayesian analysis: Provides a probability that a specific treatment is superior for that individual patient.
⭐ Results from an N-of-1 trial provide the highest level of evidence for treatment decisions in an individual patient, but are not generalizable.
High‑Yield Points - ⚡ Biggest Takeaways
- In an N-of-1 trial, a single patient serves as their own control.
- It functions as a multiple crossover study within one individual, alternating between treatment and placebo.
- Ideal for assessing treatments for chronic, stable conditions where the effect is rapid and reversible.
- The main goal is to optimize treatment for the individual patient, not to generalize findings.
- Washout periods are crucial to prevent carryover effects between treatment phases.
- Can and should be double-blinded to reduce bias.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

