Primary vs Secondary Outcomes - The Main Event
| Feature | Primary Outcome | Secondary Outcome |
|---|---|---|
| Purpose | The main question the study is designed to answer; the focus of hypothesis testing. | Additional outcomes of interest that are also observed. |
| Specification | Must be pre-specified in the study protocol before data collection begins. | Usually pre-specified, but may be exploratory; not the main focus. |
| Power | The study's sample size is calculated based on the power to detect a difference in this outcome. | The study is not typically powered to detect differences for these outcomes. |
| Example | A composite endpoint like "all-cause mortality or hospitalization" in an RCT. | Individual components of the primary endpoint, or other effects like "quality of life". |
The Primary Outcome - Planning for Power
📌 Primary outcome determines the study's Power and Purpose.
- Primary Outcome: The single, pre-specified endpoint used to determine the study's success and calculate the required sample size. It must be clinically meaningful.
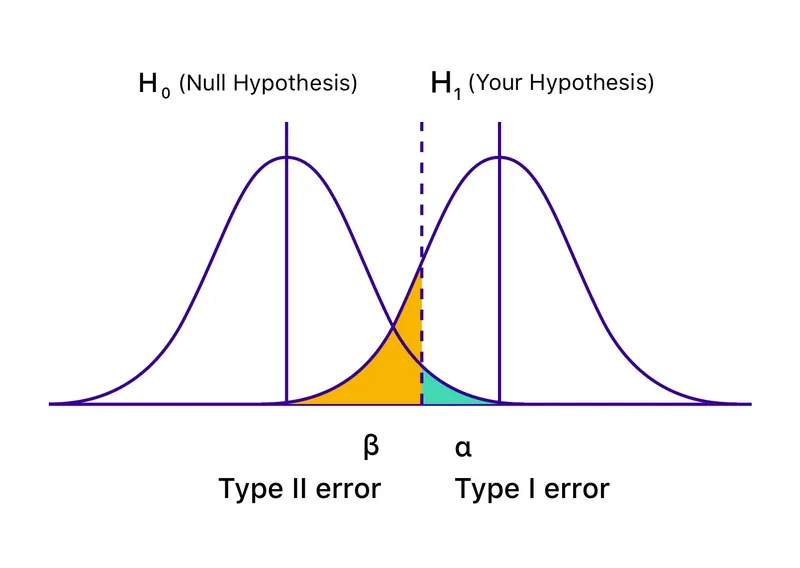
- Statistical Power: The probability of finding a true effect, if one exists. It avoids a Type II error.
- Formula: $Power = 1 - \beta$
- Standard goal: Power is typically set at ≥80%.
- Errors in Hypothesis Testing:
- Type I Error (α): False positive. Rejecting a true null hypothesis. The significance threshold (p-value) is usually <0.05.
- Type II Error (β): False negative. Failing to reject a false null hypothesis (i.e., missing a real effect).
- Endpoints:
- Single: Measures one specific outcome (e.g., all-cause mortality).
- Composite: Combines multiple endpoints (e.g., MACE: CV death, MI, stroke). ↑ statistical power by ↑ event rate.
⭐ A study with a non-significant primary outcome is considered 'negative,' even if multiple secondary outcomes are statistically significant.
Secondary & Exploratory Outcomes - Handle with Care
-
Secondary Outcomes:
- Pre-specified outcomes that are not the primary endpoint of the study.
- Often lack sufficient statistical power for definitive conclusions.
- Results are considered hypothesis-generating, not confirmatory.
- 📌 Secondary results are Suggestive and need Separate studies for confirmation.
-
Exploratory & Post-Hoc Analyses:
- Analyses that were not pre-specified in the trial protocol.
- Carry a high risk of finding spurious associations due to "p-hacking" or data dredging.
-
Multiplicity & Type I Error Inflation:
- Testing multiple outcomes increases the probability of finding a statistically significant result purely by chance.
- This is the problem of multiple comparisons, which inflates the overall Type I error rate.
⭐ Testing multiple secondary outcomes increases the risk of finding a 'significant' result by chance alone (Type I error inflation), a problem known as multiplicity.
High-Yield Points - ⚡ Biggest Takeaways
- The primary outcome is the single, pre-defined endpoint used to determine a study's success, directly testing its main hypothesis.
- Statistical power and sample size are calculated based on the primary outcome.
- Secondary outcomes are additional endpoints that are exploratory and can generate new hypotheses.
- A trial's main conclusion rests solely on the primary outcome's results.
- Finding significance in secondary outcomes alone can be a result of p-hacking or data dredging.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

