Interim Analyses - The Planned Peek
- Planned analysis of data in an ongoing RCT before the study's conclusion.
- Primary goal: To determine if the trial should be stopped early for:
- Overwhelming efficacy
- Futility (unlikely to show benefit)
- Unexpected harm
- ⚠️ Problem: Each "peek" at the data increases the overall Type I error rate (false positive) if not adjusted.
- Solution: Use a pre-specified statistical "stopping rule" with alpha-spending functions to maintain the overall study-wide $α$ (usually < 0.05).
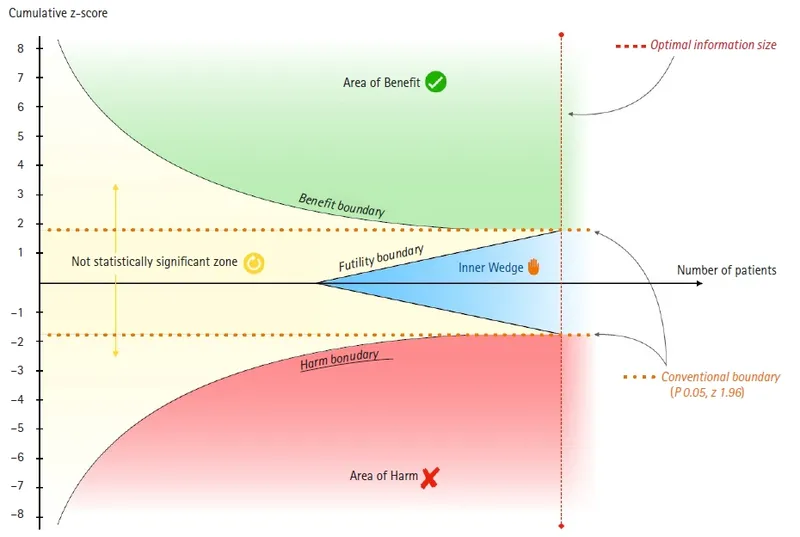
⭐ The O'Brien-Fleming boundary is a common, conservative method that requires a very low p-value for early stopping, preserving statistical power for the final analysis.
Stopping Boundaries - Guarding the Alpha
Repeatedly analyzing data during a trial inflates the overall Type I error rate (family-wise error rate), increasing the chance of a false-positive result. Stopping boundaries are pre-specified rules to control this risk.
- Formal Methods to Adjust Significance:
- O'Brien-Fleming (OBF):
- Very conservative early in the trial; requires an extremely low p-value to stop.
- The significance boundary becomes less stringent as more data is collected.
- Preserves statistical power and the final analysis uses an alpha level very close to the nominal 0.05.
- 📌 O'Brien-Fleming = Oh Boy, Far to go! (Hard to stop early).
- Pocock:
- Uses the same, constant significance level (e.g., p < 0.015) at each interim analysis.
- "Spends" alpha equally at each look, making it easier to stop early compared to OBF.
- O'Brien-Fleming (OBF):
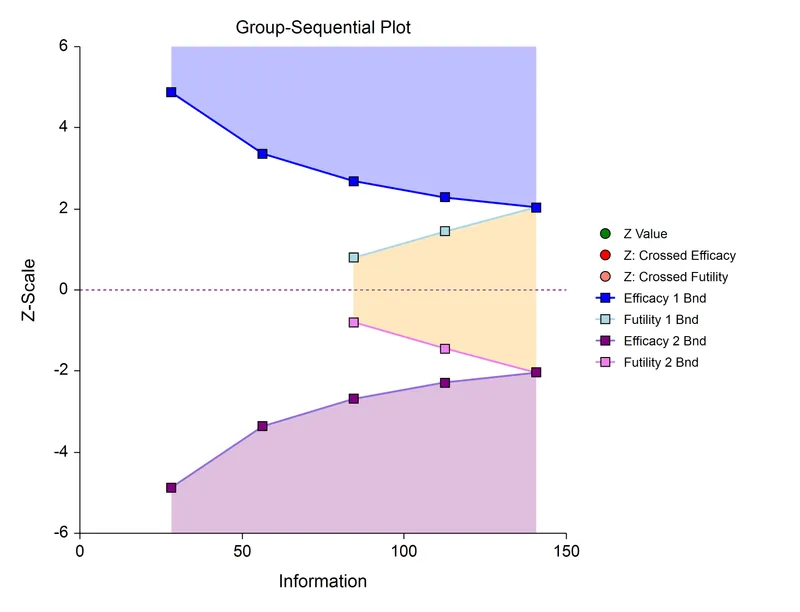
⭐ The O'Brien-Fleming method is most common for clinical trials because its early conservatism prevents premature termination based on transient effects, preserving overall trial power and integrity.
Trial Adjustments - The Ripple Effect
- Interim analyses risk ↑ Type I error (false positives) from repeated data "peeking." The total trial alpha (e.g., 0.05) must be "spent" across looks.
- Alpha-spending functions adjust significance boundaries:
- O'Brien-Fleming: Very conservative early on; preserves power.
- Pocock: Constant p-value boundary for each look.
- Haybittle-Peto Rule: Use a strict interim $p < \textbf{0.001}$ and the original final $p < \textbf{0.05}$.
- Other changes (e.g., sample size re-estimation) can be made but risk introducing bias.
⭐ Each data "peek" is another roll of the dice, increasing the cumulative chance of a false-positive (Type I error) if the significance threshold isn't adjusted.
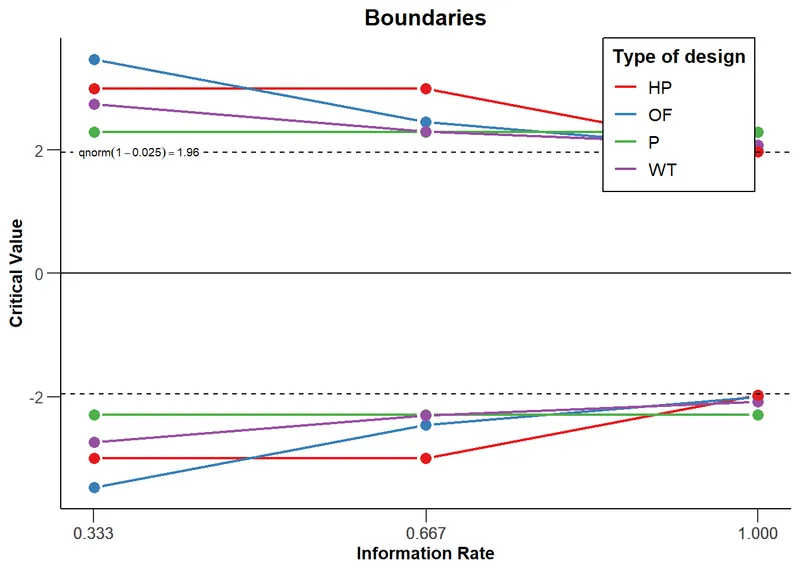
High‑Yield Points - ⚡ Biggest Takeaways
- Interim analyses are performed during an RCT to monitor for efficacy, futility, or harm.
- Requires an independent Data and Safety Monitoring Board (DSMB) to review unblinded data.
- Uses pre-specified stopping boundaries to guide decisions on trial continuation or termination.
- Multiple analyses increase the risk of Type I error (false positives).
- Statistical adjustments, like alpha-spending functions, are used to maintain the overall p-value < 0.05.
- Early stopping for benefit is a major ethical consideration.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

