Energy Requirements - Paying the ATP Toll
- The synthesis of one molecule of urea is an energy-intensive process, requiring the equivalent of 4 high-energy phosphate bonds derived from 3 ATP molecules.
- ATP Consumption Steps:
- Carbamoyl Phosphate Synthetase I (Mitochondria): Consumes 2 ATP.
- Argininosuccinate Synthetase (Cytosol): Consumes 1 ATP, which is hydrolyzed to AMP and pyrophosphate (PPi).
⭐ The hydrolysis of pyrophosphate (PPi → 2 Pi) is energetically equivalent to breaking a high-energy phosphate bond. This brings the total energetic cost to 4 ATP equivalents, making the urea cycle effectively irreversible.
- Overall Equation:
- $2 ext{ NH}_4^+ + ext{HCO}_3^- + 3 ext{ ATP} ightarrow ext{Urea} + 2 ext{ ADP} + ext{AMP} + 2 ext{ P}_i + ext{PP}_i$
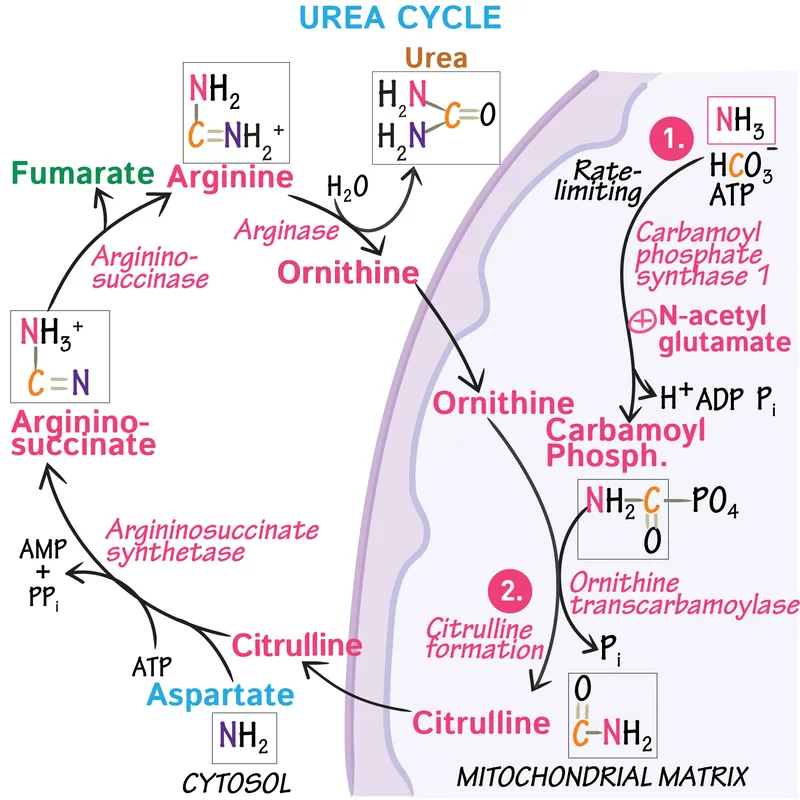
ATP-Consuming Steps - Where the Energy Goes
The urea cycle consumes a total of 3 ATP molecules, equivalent to 4 high-energy phosphate bonds, to synthesize one molecule of urea.
-
Step 1: Carbamoyl Phosphate Synthetase I (CPS-I)
- Location: Mitochondria
- Action: Catalyzes the condensation of $NH_4^+$ and $HCO_3^-$.
- Cost: Consumes 2 ATP molecules.
- Reaction: $NH_4^+ + HCO_3^- + 2ATP \rightarrow$ Carbamoyl Phosphate $+ 2ADP + P_i$
-
Step 2: Argininosuccinate Synthetase
- Location: Cytosol
- Action: Links citrulline with aspartate.
- Cost: Consumes 1 ATP, which is hydrolyzed to AMP and pyrophosphate ($PP_i$). The subsequent hydrolysis of $PP_i$ to $2P_i$ makes this step energetically equivalent to using 2 ATP.
- Reaction: Citrulline + Aspartate + ATP $\rightarrow$ Argininosuccinate $+ AMP + PP_i$
⭐ High-Yield Fact: The net energy cost of the urea cycle is 4 high-energy phosphate bonds ($2$ from ATP $\rightarrow$ ADP at the CPS-I step, and $2$ from ATP $\rightarrow$ AMP at the argininosuccinate synthetase step). This is a frequent point of confusion on exams.
Net Energy Cost - The Final Bill
-
Gross Cost: 4 high-energy phosphate bonds are consumed to synthesize one molecule of urea.
- Carbamoyl Phosphate Synthetase I (CPS-I): Consumes 2 ATP.
- Argininosuccinate Synthetase: Consumes 1 ATP, but hydrolyzes it to AMP and pyrophosphate (PPi). The subsequent hydrolysis of PPi makes this step equivalent to spending 2 ATP.
-
Overall Reaction:
- $Aspartate + NH_3 + HCO_3^- + 3 ATP \rightarrow Urea + Fumarate + 2 ADP + AMP + 2 P_i + PP_i$
⭐ The Krebs Bicycle Connection: The cycle isn't isolated. Fumarate re-enters the mitochondria and is converted to malate, generating 1 NADH. This NADH produces ~2.5 ATP via the ETC, effectively lowering the net cost of urea synthesis to ~1.5 ATP.
High‑Yield Points - ⚡ Biggest Takeaways
- The urea cycle consumes 3 ATP to synthesize one molecule of urea.
- A total of 4 high-energy phosphate bonds are cleaved: 2 ATP → 2 ADP + 2 Pi, and 1 ATP → AMP + PPi.
- The rate-limiting step, catalyzed by Carbamoyl Phosphate Synthetase I (CPS I), requires 2 ATP.
- The Argininosuccinate Synthetase step consumes the third ATP.
- Overall, the urea cycle is a highly energy-expensive, irreversible pathway essential for ammonia detoxification.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

