RTK Fundamentals - The Dimer's Dance
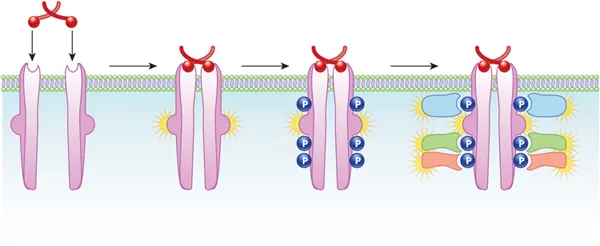
- Structure: Monomeric transmembrane proteins with an extracellular ligand-binding domain and an intracellular tyrosine kinase domain.
- Activation: Triggered by the binding of a specific ligand (e.g., insulin, EGF, PDGF).
- Dimerization: Ligand binding causes two receptor monomers to form a dimer.
- Autophosphorylation: The kinase domain of one monomer phosphorylates tyrosine residues on the tail of the other, and vice-versa (cross-phosphorylation).
- Docking & Signaling: Phosphorylated tyrosines serve as docking sites for signaling proteins with SH2 domains.
⭐ Many oncogenes, like HER2, are RTKs. Overexpression or mutation can lead to ligand-independent dimerization and constant activation, driving uncontrolled cell growth.
MAP Kinase Pathway - Growth Signal Freeway
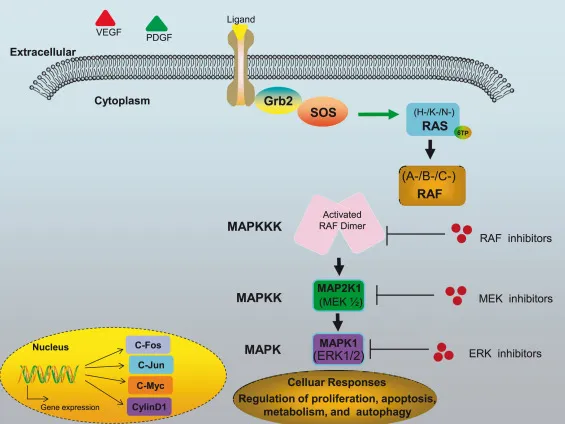
- Initiation: Growth factor (e.g., EGF, FGF) binds to its specific Receptor Tyrosine Kinase (RTK), triggering receptor dimerization and autophosphorylation.
- Transduction: The adaptor protein GRB2 and Guanine nucleotide Exchange Factor SOS are recruited to the phosphorylated RTK.
- Activation: SOS promotes the exchange of GDP for GTP on Ras, a small G-protein. This activates Ras.
- Cascade: Activated Ras-GTP initiates a phosphorylation cascade.
⭐ Ras is a key proto-oncogene. Activating mutations, common in pancreatic, lung, and colon cancers, lock Ras in a GTP-bound (active) state, driving uncontrolled proliferation even without growth factors.
PI3K/Akt Pathway - Survival & Growth Pro
- Activation: Ligand binding to RTK recruits PI3K (Phosphoinositide 3-kinase).
- Mechanism: PI3K phosphorylates membrane lipid PIP2 to form PIP3.
- PIP3 acts as a docking site, leading to the activation of Akt (Protein Kinase B).
- Downstream Effects: Akt activation promotes:
- Cell Survival: ↓ Apoptosis by inhibiting proteins like Bad and FOXO.
- Cell Growth: Activates mTOR, boosting protein synthesis.
- Metabolism: ↑ Glucose uptake (GLUT4) & glycogen synthesis.
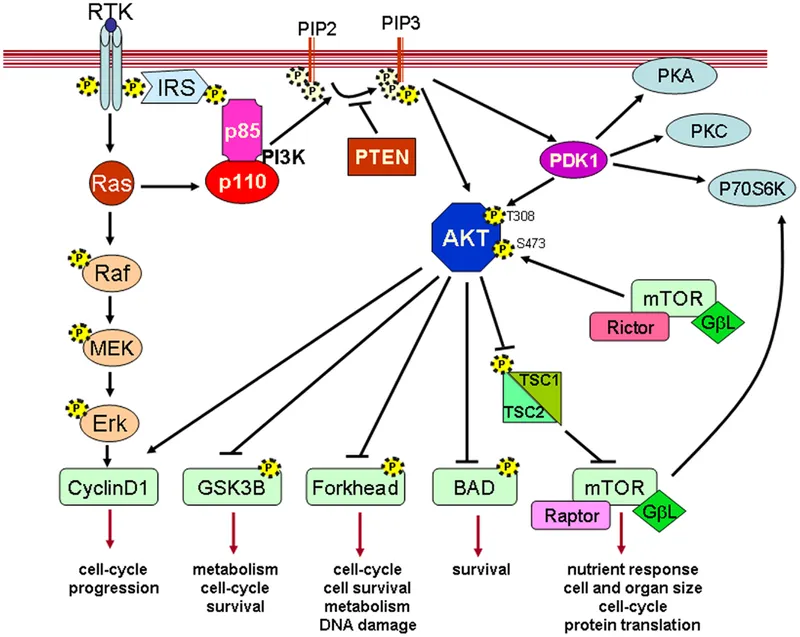
⭐ PTEN (Phosphatase and Tensin Homolog) is a key tumor suppressor that dephosphorylates PIP3, turning off this pro-survival pathway. Loss-of-function mutations are common in cancers like endometrial, prostate, and glioblastoma.
Insulin Receptor - The Glucose Gatekeeper
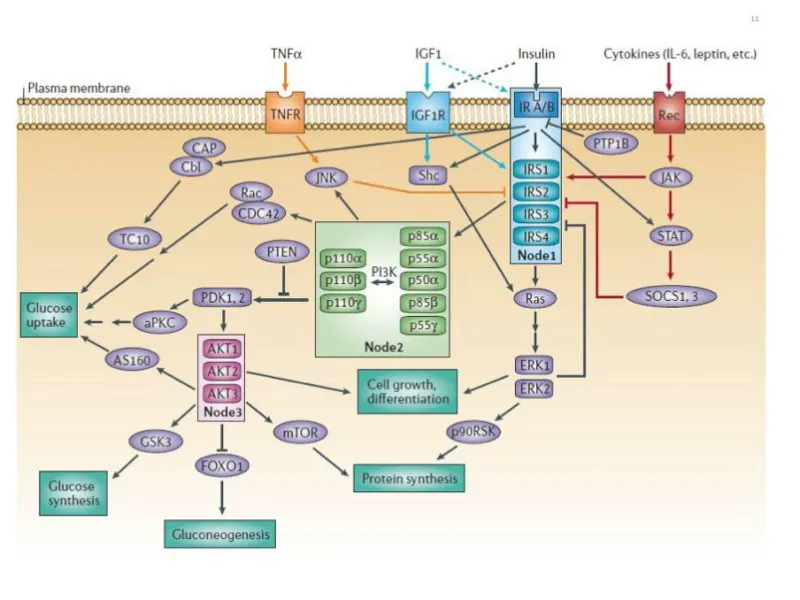
- Structure: Pre-formed dimer with intrinsic tyrosine kinase activity (RTK).
- Mechanism: Insulin binding → autophosphorylation of tyrosine residues.
- Recruits Insulin Receptor Substrate (IRS-1).
- Dual Pathways Activated by IRS-1:
- PI3K Pathway (Metabolic): IRS-1 → PI3K → PIP3 → Akt (PKB) activation.
- Promotes translocation of GLUT4 transporters to the cell surface in muscle & adipose tissue.
- ↑ Glucose uptake.
- MAPK Pathway (Mitogenic): IRS-1 → GRB2 → Ras → MAPK activation.
- Regulates gene expression for cell growth & proliferation.
- PI3K Pathway (Metabolic): IRS-1 → PI3K → PIP3 → Akt (PKB) activation.
⭐ High-Yield: Insulin signaling has two arms. The PI3K/Akt pathway controls metabolic actions (glucose uptake), while the RAS/MAPK pathway controls mitogenic effects (cell growth). This separation is key in understanding insulin resistance pathologies.
High‑Yield Points - ⚡ Biggest Takeaways
- Ligand binding triggers receptor dimerization and subsequent autophosphorylation of tyrosine residues.
- Phosphorylated tyrosines serve as docking sites for signaling proteins containing SH2 domains.
- Key downstream cascades are the MAP kinase pathway (RAS-RAF-MEK-ERK) and the PI3K/AKT pathway.
- The insulin receptor is a classic example, though it exists as a pre-formed dimer.
- Primarily bind growth factors like EGF, PDGF, and FGF, regulating cell growth and proliferation.
- Gain-of-function mutations are a hallmark of many cancers, making them key oncologic drug targets.
Unlock the full lesson and continue reading
Signup to continue reading this lesson and unlimited access questions, flashcards, AI notes, and more

